Government to Focus Support on Developing First Domestic COVID-19 Vaccine… 262.7 Billion Won Budget Allocated This Year
Strategic Support for Clinical Phase 3 Treatment Drugs... Budget Increased by 20% Compared to Last Year
[Asia Economy Reporter Seo So-jeong] The government will focus support on items nearing entry into Phase 3 clinical trials to accelerate the development of COVID-19 treatments. It plans to provide strategic support through a portfolio development strategy based on severity, ranging from mild to severe cases.
Additionally, next year, to enable the first domestic vaccine inoculation, the government will select items with potential for clinical progress, such as synthetic antigen vaccines, as priority items for concentrated support, considering development trends.
On the 19th, the government held the 9th meeting of the Pan-Governmental Support Committee for COVID-19 Treatment and Vaccine Development to discuss this year's support strategies for COVID-19 treatments and vaccines, the development status of mobile negative pressure wards, and plans for early commercialization.
To alleviate the biggest challenge faced by companies in developing domestic treatments and vaccines?the burden of clinical trial costs?the government will invest about 130 billion KRW this year and promote support through funds and other means.
To resolve difficulties in recruiting clinical trial participants, the government will actively support companies' overseas clinical trials through the Overseas Clinical Trial Comprehensive Consultation Center (National Clinical Trial Foundation), including clinical design and strategy formulation.
The government will strengthen international cooperation to rapidly establish and utilize immune correlates of protection (ICP) for analyzing the correlation between immunogenicity (antibody levels, duration, etc.) induced by vaccines and protective effects, and for comparison with vaccines proven effective through surrogate markers.
Furthermore, the number of National Infectious Disease Clinical Trial Centers will be expanded from the existing three to five consortia this year, and related systems and infrastructure will be enhanced by revising the Pharmaceutical Affairs Act to introduce a Central Institutional Review Board.
To support clinical trials, the government will promote the establishment of a public vaccine clinical trial cluster and train specialized personnel for each clinical stage from preclinical to commercialization.
From the first half of this year, the government will conduct nationally led rapid clinical research to ensure treatments can be quickly used for patient care during infectious disease crises, actively utilizing treatment-use approval systems and investigator-initiated clinical studies.
Upon completion of treatment development, the Ministry of Food and Drug Safety will expand dedicated teams to ensure rapid approval and national batch release, while continuously monitoring overseas trends in excellent treatments and vaccines and considering procurement through expert consultation if necessary.
In addition, to prepare for new infectious diseases, investment will be expanded in next-generation treatment and vaccine platform technologies, with active efforts in mRNA vaccine technology development, including operating a messenger ribonucleic acid (mRNA) vaccine project group in the form of a technology convergence consortium.
Moreover, the government will promote development by supporting matching and linkage cooperation between development and manufacturing companies, and expand global cooperation for early technology acquisition through Korea-US joint research, joint research between global pharmaceutical companies and domestic firms, and contract manufacturing.
Rapid Support for Domestic Vaccine Development Strategy... Early Commercialization of Mobile Negative Pressure Wards
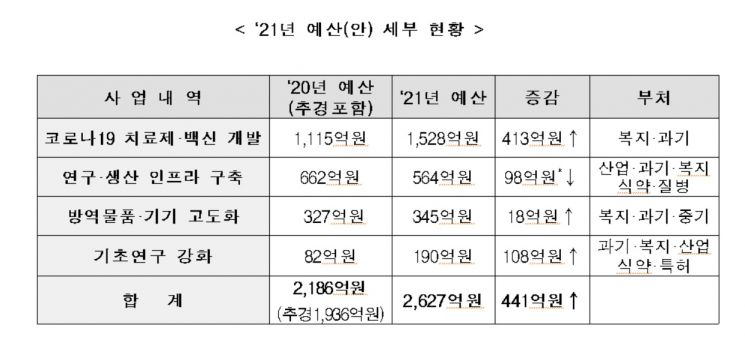
The government plans to support a total budget of 262.7 billion KRW this year for COVID-19 treatment and vaccine development, a 44.1 billion KRW (20%) increase compared to last year.
First, 152.8 billion KRW will be invested to strengthen research support in clinical and non-clinical stages to ensure the success of COVID-19 treatment and vaccine development and clinical trials.
564 billion KRW will be allocated for the development of testing methods for treatments and vaccines, and for establishing experimental facilities and equipment to build production infrastructure. A budget of 34.5 billion KRW will be invested in the development, advancement, and localization of next-generation infectious disease equipment and devices such as rapid diagnostics and intelligent devices.
To strengthen basic research such as core infectious disease technology development and evaluation research for approval support, 19 billion KRW will be provided.
The government will promote early commercialization of mobile negative pressure wards capable of treating moderate to severe infectious disease patients through cooperation among related ministries.
Hot Picks Today
 "Now Our Salaries Are 10 Million Won a Month" Record High... Semiconductor Boom Drives Performance Bonuses at Major Electronic Component Firms
"Now Our Salaries Are 10 Million Won a Month" Record High... Semiconductor Boom Drives Performance Bonuses at Major Electronic Component Firms
- Wallets Open Wide on Big News...300 Trillion Won Heads to the U.S., "Tax Breaks" Fail to Keep Funds at Home
- "Hope You Enjoy the 'Welfare' for Bereavement of Children"... Ridicule of Strike Non-Participants Intensifies Union Conflict at Samsung Electronics
- "Heading for 2 Million Won": The Company the Securities Industry Says Not to Doubt [Weekend Money]
- Experts Already Watching Closely..."Target Price Set at 970,000 Won" Only Upward Momentum Remains [Weekend Money]
Kwon Deok-cheol, Minister of Health and Welfare, stated, "Even as COVID-19 vaccinations begin in earnest, we must secure our own capabilities to prepare for new infectious diseases in the long term. Starting with the development of domestic treatments, we will gather pan-governmental capabilities to develop the first vaccine."
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
![[Image source=Yonhap News]](https://cphoto.asiae.co.kr/listimglink/1/2021020416572162900_1612425442.jpg)