Government Finalizes Moderna Vaccine Purchase Contract... Total Supply Secured for 56 Million People (Comprehensive)
KDCA to Introduce 20 Million Moderna Vaccine Doses in Q2 Next Year
Final Step After Covax, AstraZeneca, Pfizer, Janssen
"Supply Risk Diversification... Sequential Vaccination Starting Q1 Next Year"
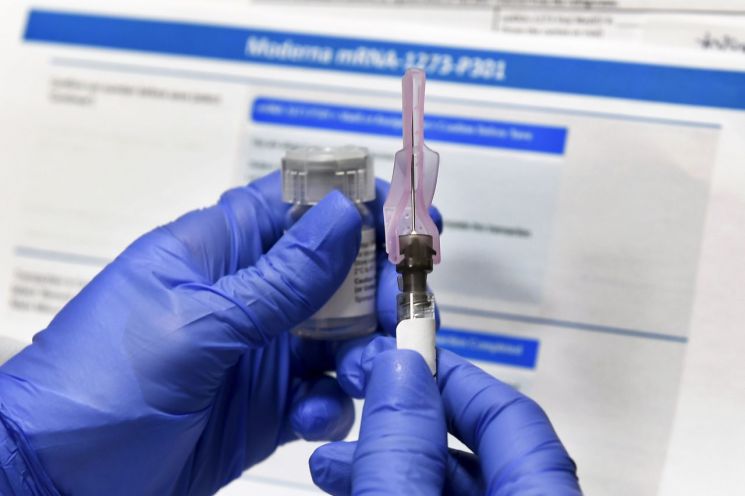
[Asia Economy Reporters Choi Dae-yeol and Kim Heung-soon] The government announced on the 31st that it has signed a pre-purchase contract for the Moderna COVID-19 vaccine. With this contract, all agreements for the 56 million doses of vaccines secured by our government have been finalized.
This contract follows the agreement reached on the 28th between President Moon Jae-in and Moderna CEO St?phane Bancel, after which the contract wording was negotiated. According to this contract, our government will receive 40 million doses of the Moderna vaccine (one dose per shot, enough for 20 million people) by the second quarter. This vaccine requires two doses administered at intervals.
Initially, our government planned to purchase about 20 million doses from this company, but the amount was doubled. Following Pfizer-BioNTech, Moderna developed the second vaccine worldwide using a new technology called messenger RNA. Since it must be distributed under ultra-low temperature conditions and storage is complicated, it is expected to take considerable time before widespread vaccination can be conducted.

Jung Eun-kyung, Director of the Central Disease Control Headquarters (Director of the Korea Disease Control and Prevention Agency)
Securing 56 Million Vaccine Doses for Domestic Supply
Enough for Vaccinating the Entire Population
With the completion of the contract with Moderna, all supply contracts have been finalized. Starting with 20 million doses through the joint development and purchase project COVAX Facility, contracts for 20 million doses of AstraZeneca, 20 million doses of Pfizer, and 6 million doses of Janssen have been completed, totaling 106 million doses of vaccines purchased.
The Janssen vaccine requires only one dose, while the others require two doses. In terms of population, this equates to 56 million doses, enough to vaccinate the entire Korean population. However, the specific manufacturer of the vaccines through the COVAX Facility has not been confirmed, and since the Janssen vaccine has not yet completed final development, the actual quantities arriving domestically may vary.
The government was reportedly considering importing vaccines from domestic companies with which it had contract manufacturing and development agreements, such as Novavax or Pfizer, but at a briefing on the same day, Jeong Eun-kyung, Director of the Korea Disease Control and Prevention Agency, stated that there are currently no plans to consider additional purchases.
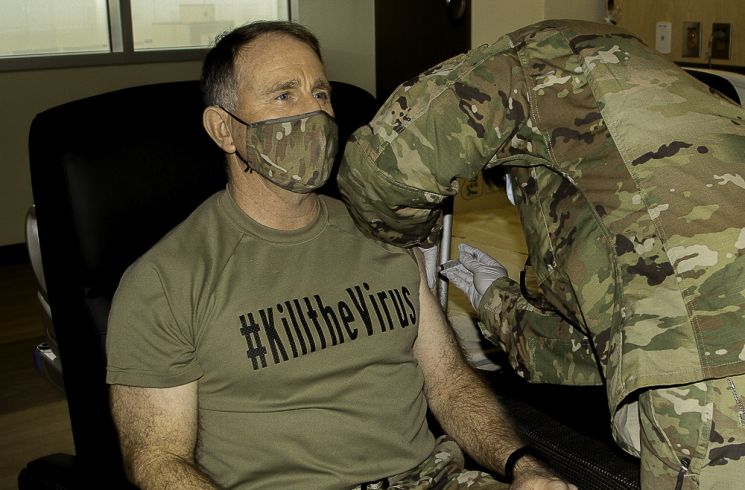
On the 29th (Korean time), Robert Abrams, Commander of the ROK-U.S. Combined Forces Command and Commander of United States Forces Korea, received a COVID-19 vaccination at Brian Allgood Army Community Hospital on the Humphreys U.S. Army Garrison in Pyeongtaek, Gyeonggi Province.
Among the pre-purchased vaccines negotiated individually with manufacturers, AstraZeneca is expected to be the earliest, anticipated in the first quarter of next year. Janssen and Moderna are scheduled for the second quarter, and Pfizer for the third quarter. These schedules may change due to issues in the actual production and manufacturing processes. Considering the ongoing COVID-19 outbreak domestically, the government is also negotiating with each manufacturer to advance the supply schedule.
Director Jeong said, "We pursued vaccine purchases by preparing a portfolio that diversifies risk by using different manufacturing methods and manufacturers. The vaccine contracts have been completed, and we will do our best to prepare detailed implementation plans so that vaccinations can proceed smoothly, quickly, and safely starting next year."
Hot Picks Today
!["Stock Set to Double: This Company Smiles Every Time a Data Center Is Built [Click e-Stock]"](https://cwcontent.asiae.co.kr/asiaresize/93/2026050416112750184_1777878687.jpg) "Stock Set to Double: This Company Smiles Every...
"Stock Set to Double: This Company Smiles Every...
- "Is Yours Just Gathering Dust at Home? Millennials & Gen Z Rediscover Digicams O...
- "Continuous Groundwater Pumping Causes Mexico City to Sink 24cm Annually... 'Gia...
- "I Take Full Responsibility"... Seongjae Ahn Issues Direct Apology for 'Wine Swi...
- “She Shouted, ‘The Rope Isn’t Tied!’... Chinese Woman Falls from 168m Cliff ...
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.