MFDS: "Results on Effectiveness and Safety of White Particle Flu Vaccine to be Released Next Week"
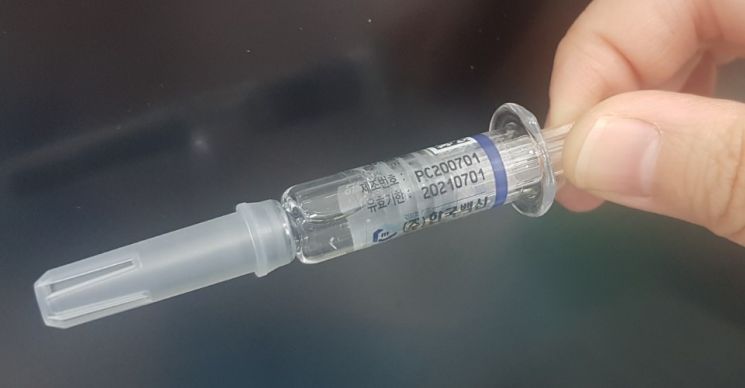
The influenza vaccine 'Kovax Flu Quadrivalent PF' by Korea Vaccine Co., Ltd., in which white particles were found. The company has recalled a total of 615,000 doses from four manufacturing batches.
[Asia Economy Reporter Choi Dae-yeol] The Ministry of Food and Drug Safety plans to disclose the test results of the influenza (flu) vaccine in which white particles were found as early as next week. Previously, Korea Vaccine, the manufacturer of this vaccine, voluntarily decided to recall it and has collected most of the doses.
On the 23rd, the Ministry of Food and Drug Safety stated, "We are conducting tests to verify the efficacy and effectiveness of the vaccine with white particles, the protein characteristics of the white particles, and safety in animal tests," adding, "The test results are expected to be released next week."
Earlier, Lee Eui-kyung, the head of the Ministry of Food and Drug Safety, testified at the National Assembly audit the day before, stating that experiments are being conducted to determine the cause of the white particles and their effects. After reviewing the report received earlier this month, it was initially concluded that the particles were protein aggregates and posed no safety issues. However, since the particles could block the injection needle, the manufacturer decided to recall the vaccine voluntarily.
Prior to this, some flu vaccines that were not properly temperature-controlled during transportation were found to have no quality issues after inspection. Nevertheless, to dispel concerns about efficacy and safety, the government decided to recall most of the vaccines.
Hot Picks Today
 "Suspicious Timing?"...Trump Traded Stocks After Praising Wartime Capabilities
"Suspicious Timing?"...Trump Traded Stocks After Praising Wartime Capabilities
- "I Went to 10 Convenience Stores and Still Couldn't Buy It": The Bread Sensation That Sold 100 Million Units Already [The Way We Shop Now]
- Trump Pressures Taiwan to Relocate Semiconductor Industry to U.S., Says "Taiwan Took Away America's Semiconductor Industry"
- There Is a Distinct Age When Physical Abilities Decline Rapidly... From What Age Do Strength and Endurance Drop?
- "Contact Me First If Houses Are Built": Wealthy Clients Eyeing... Will Ultra-High-End Residences Worth 20 Billion Won Be Developed? [Real Estate AtoZ]
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.







