How Did the Meticulously Planned Flu Vaccination Schedule Fall Apart?
Health Authorities Greatly Expand Free Vaccination Eligibility
Guidelines Cover Purchase, Distribution, Vaccination, and Redistribution
But Frontline Sites Struggle to Implement Them Properly

A notice for free flu vaccinations is posted at a hospital in downtown Seoul. Photo by Kim Hyun-min kimhyun81@
View original image[Asia Economy Reporter Choi Dae-yeol] Jung Eun-kyung, the Commissioner of the Korea Disease Control and Prevention Agency (KDCA), who oversees the national vaccination plan, put considerable effort into devising this year’s influenza (flu) vaccination plan. Since the symptoms are similar to those of COVID-19, she believed that suppressing the outbreak as much as possible would prevent confusion at frontline medical institutions. Initially, the free vaccination target age was planned to increase by only one year from under 12 last year to 13 this year, but considering the high level of group activities, the target was significantly raised to 18 years old in the supplementary budget for this year.
This alone increased the vaccination target by nearly 3 million people. The free vaccination target for the elderly was also lowered from 65 years old to 62. The target vaccination rate was set higher than last year, and the timing for elderly vaccinations was further subdivided. To ensure safe vaccinations, the previous recommendation of 'a maximum of 100 vaccinations per doctor' was made enforceable this year, allowing contract termination if violated. Additionally, vaccinations were made possible at social welfare facilities with contracted doctors to widely cover vulnerable groups.
Although there were some complaints from vaccine manufacturers and importers, the government succeeded in purchasing vaccines at a low price. They bought 12.59 million doses, equivalent to one in every four Koreans, at less than 10,000 KRW per dose. Considering that the market price for quadrivalent vaccines is around 16,000 KRW, this was an efficient purchase within a limited budget. Anticipating regional differences in vaccination rates during the vaccination period, detailed redistribution plans among local health centers, suppliers, and entrusted medical institutions were prepared. A system was also established for joint monitoring by the Ministry of Food and Drug Safety, responsible for national batch approval, and the Health Insurance Review and Assessment Service, which can track usage in real time.
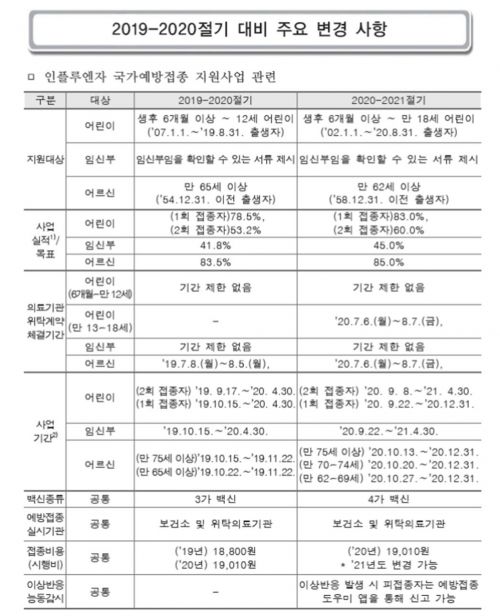
Part of the Influenza National Vaccination Support Project guidelines distributed by the Korea Disease Control and Prevention Agency (currently the Korea Disease Control and Prevention Administration) for reference by frontline medical institutions. It clearly explains the changes between last year and this year.
View original imageReports Received of Suspected Room Temperature Exposure in Some Areas
If No Reports, Authorities Might Not Have Detected It
Despite these thorough preparations, a problem occurred. There were suspicions that vaccines were exposed to room temperature during transportation in some areas. This indicates that no matter how perfect the guidelines are, it is not easy for them to be fully implemented at the frontline. The possibility of room temperature exposure during transportation, which became an issue, is a matter that should have been anticipated for biopharmaceuticals. Although there were guidelines to comply with cold chain distribution, it is presumed that some areas failed to fully adhere to them.
The KDCA (formerly the Korea Centers for Disease Control and Prevention) creates management guidelines every year covering all areas from vaccine procurement to transportation, distribution, and post-management, distributing them to all institutions involved in the vaccination program. The guidelines exceed 200 pages and contain detailed tasks that local governments, health centers, and entrusted medical institutions must perform.
However, it is also true that there are physical limits to managing and supervising whether these guidelines are properly followed at each site. The suspicion of room temperature exposure of flu vaccines was first detected by the authorities through a report from someone. Without such a report, it is highly likely that the issue would have gone unnoticed.

Jung Eun-kyung, Commissioner of the Korea Disease Control and Prevention Agency <이미지:Yonhap News>
View original image2303 Additional Vaccinations Administered Amid Investigation of Suspected Room Temperature Exposure
Vaccination Timing Not Followed, Gaps in Free and Paid Vaccine Management
Authorities: "We Will Review Improvement Measures for the National Vaccination Support Program"
Issues such as failure to adhere to vaccination timing or mixing management of free and paid vaccines would have been difficult to uncover if this incident had not occurred. According to the KDCA, as of the 2nd, 2,303 people had been vaccinated with vaccines under investigation for suspected room temperature exposure. Considering that the government initially stated on the 22nd, when the national vaccination was temporarily suspended, that no one had been vaccinated with the vaccines under investigation, this clearly reveals weaknesses in the management system.
During the investigation of government-procured vaccine administration, it was revealed that free vaccines were being administered to individuals not eligible for free vaccination, and some elderly vaccinations scheduled for mid-month were conducted nearly a month earlier. The institutions found violating guidelines are likely to face only contract termination from entrusted medical institutions as punishment.
The government spends 150 billion KRW on vaccines alone for the free flu vaccination program, and when including vaccination costs, the total reaches 500 billion KRW. Adding paid vaccinations for the general adult population, the amount is even larger. Considering that flu vaccination targets involve about three out of five citizens, encompassing more than half the population directly or indirectly, this is a national health project that requires guidelines ensuring more reliable enforcement.
The investigation into vaccines suspected of room temperature exposure is expected to conclude within this week. In addition to the 5.78 million doses in question, the transportation of the remaining 7 million doses is also being inspected. The KDCA stated, "Cases of vaccination due to management negligence or non-compliance with guidelines are being identified during the investigation," and added, "We plan to review improvement measures for the national influenza vaccination support program, including these cases."
Hot Picks Today
 "Now Our Salaries Are 10 Million Won a Month" Record High... Semiconductor Boom Drives Performance Bonuses at Major Electronic Component Firms
"Now Our Salaries Are 10 Million Won a Month" Record High... Semiconductor Boom Drives Performance Bonuses at Major Electronic Component Firms
- Wallets Open Wide on Big News...300 Trillion Won Heads to the U.S., "Tax Breaks" Fail to Keep Funds at Home
- "Hope You Enjoy the 'Welfare' for Bereavement of Children"... Ridicule of Strike Non-Participants Intensifies Union Conflict at Samsung Electronics
- President Lee Praises 'Everyone's Startup' as Applications Surpass 62,000
- Experts Already Watching Closely..."Target Price Set at 970,000 Won" Only Upward Momentum Remains [Weekend Money]
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.