Trump: "FDA Grants Emergency Use Authorization for Remdesivir" (Comprehensive)
[Asia Economy New York=Correspondent Baek Jong-min] Remdesivir, which has attracted attention as a treatment for the novel coronavirus infection (COVID-19), has received emergency use authorization from the U.S. Food and Drug Administration (FDA).
U.S. President Donald Trump announced this on the 1st (local time) at the White House Oval Office together with Daniel O'Day, CEO of Gilead. President Trump added that remdesivir will be distributed to hospitals starting from the 4th.
O'Day, CEO of Gilead, the manufacturer of remdesivir, also announced that they will donate enough remdesivir for 1.5 million people.
Remdesivir was originally developed as an Ebola treatment, but it rapidly emerged as a treatment after showing effectiveness in clinical trials for COVID-19.
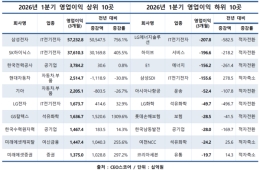
Hot Picks Today
 "Now Our Salaries Are 10 Million Won a Month" Record High... Semiconductor Boom Drives Performance Bonuses at Major Electronic Component Firms
"Now Our Salaries Are 10 Million Won a Month" Record High... Semiconductor Boom Drives Performance Bonuses at Major Electronic Component Firms
- 'Still Hesitant? If You're Wondering Whether KOSPI Will Rise, This Is the Number You Must Watch [Weekend Money]'
- "Heading for 2 Million Won": The Company the Securities Industry Says Not to Doubt [Weekend Money]
- Fire Breaks Out on 29th Floor of Tower Palace in Seoul; About 40 Residents Evacuated, No Injuries Reported
- Is It Really Like an Illness? "I Can't Wait to Go Again"—Over 1 Million Visited in Q1, Now 'Busanbyeong' Takes Hold [K-Holic]
Gilead's stock price fell 4.8% that day but rose about 2% based on the news released after the market closed.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
![[Image source=EPA Yonhap News]](https://cphoto.asiae.co.kr/listimglink/1/2020050207102616140_1588371026.jpg)