Teragen Etex "Discovering COVID-19 Drug Candidates with AI"
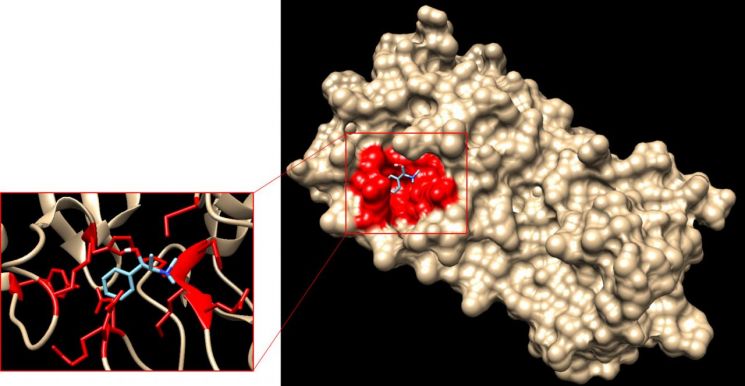
Demonstration scene of Theragen Etex's binding prediction technology used in the discovery of COVID-19 therapeutic candidate substances
View original image[Asia Economy Reporter Choi Dae-yeol] Theragen Etex announced on the 17th that it has discovered candidate substances for a novel coronavirus (COVID-19) treatment by utilizing artificial intelligence (AI) drug development platform technology and genome analysis technology.
The company stated that it recently conducted drug repositioning screening research targeting 1,880 FDA-approved drugs in the United States and identified five candidate substances for treatment. Unlike previous similar studies that mainly used the protein structures of the Severe Acute Respiratory Syndrome (SARS) or Middle East Respiratory Syndrome (MERS) viruses, the company explained that it increased accuracy and the likelihood of development success by performing its own COVID-19 genome sequencing.
Among the initially acquired candidate substances, drugs currently used by frontline medical staff as treatments, such as chloroquine, remdesivir, ribavirin, and favipiravir, ranked highly. The final candidate substances included respiratory disease treatments and cardiovascular disease treatments among the top ranks. The company anticipated that "respiratory disease treatments can be expected to have antiviral effects as well as symptom relief, and cardiovascular disease treatments could be applied to patients with underlying cardiovascular conditions."
Prior to this research, Theragen Etex analyzed the mechanism of action of the coronavirus and selected two protein targets. They plan to conduct drug efficacy tests with reputable research institutions on the candidate substances obtained this time, while also discovering additional candidate substances among 12 other target proteins. For verified drugs, they intend to apply for clinical trial approval from the Ministry of Food and Drug Safety aiming for early authorization.
Oh Yong-ho, head of the new drug development center at Theragen Etex, said, "While actively pursuing the development of a COVID-19 treatment that can be commercialized early, we will establish our own genome database for viral infectious diseases to utilize it for the development of treatments for other viruses in the future."
Hot Picks Today
!["Not Everyone Can Afford This: Inside the World of the True Top 0.1% [Luxury World]"](https://cwcontent.asiae.co.kr/asiaresize/93/2026051507261063923_1778797570.png) "Not Everyone Can Afford This: Inside the World of the True Top 0.1% [Luxury World]"
"Not Everyone Can Afford This: Inside the World of the True Top 0.1% [Luxury World]"
- "Sold Out Everywhere" The Surprising Story of the 'Purple Gold' Philippine Yam That Has Captivated the World [Delicious Stories]
- While All Eyes Were on Samsung and Hynix, This Company Surged 50% to New Highs in Four Days [Weekend Money]
- "Now Our Salaries Are 10 Million Won a Month" Record High... Semiconductor Boom Drives Performance Bonuses at Major Electronic Component Firms
- Experts Already Watching Closely..."Target Price Set at 970,000 Won" Only Upward Momentum Remains [Weekend Money]
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.







