Samsung Medical Center and InSung Medical Obtain First Domestic Approval for Cardiopulmonary Oxygenator
Essential Device for ECMO and Cardiopulmonary Bypass
First Step Toward Localization After Full Import Reliance
Stable Supply Expected Amid Supply Chain Uncertainties
The cardiopulmonary oxygenator jointly developed by Samsung Medical Center and Insung Medical has become the first domestically produced product to receive approval from the Ministry of Food and Drug Safety. This marks the first case of domestic production for an essential life-support device that had previously relied entirely on imports.
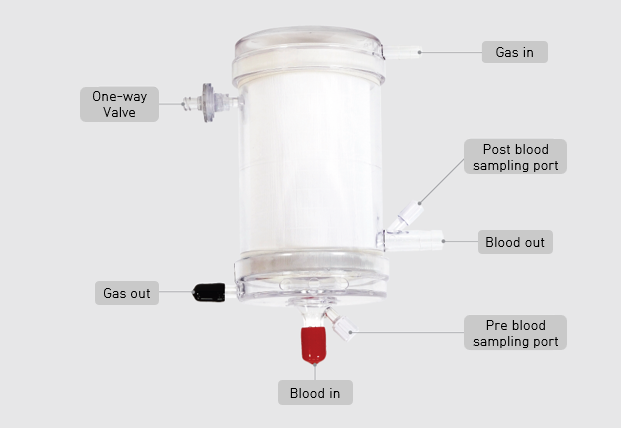
The cardiopulmonary oxygenator jointly developed by Samsung Medical Center and Insung Medical has become the first domestically produced product to receive approval from the Ministry of Food and Drug Safety. Samsung Medical Center
View original imageAccording to Samsung Medical Center on April 29, the approved product is called 'ISOx'. It serves as an artificial lung that supplies oxygen to blood and removes carbon dioxide for up to six hours during extracorporeal circulation treatments such as extracorporeal membrane oxygenation (ECMO) and cardiopulmonary bypass surgery. The development was led by Insung Medical, with Samsung Medical Center participating as a joint research institution, as part of the multi-ministerial full-cycle medical device R&D project.
Until now, all oxygenators used in Korea have been entirely dependent on imports. With ongoing instability in global supply chains raising concerns about potential supply disruptions, this approval is expected to serve as a turning point for establishing a stable supply system.
The product design directly incorporated the ECMO clinical experience of Samsung Medical Center. Since introducing modern ECMO treatment in Korea for the first time in 2003, Samsung Medical Center has accumulated over 2,500 treatment cases. The research team led by Professor Cho Yanghyun of the Department of Cardiothoracic Surgery, which spearheaded the development, integrated clinical data from the initial design stage and led both the extracorporeal performance testing and preclinical trials.
Hot Picks Today
![Home Appliance Woes Mount, Yet 45 Trillion Won in Bonuses? Samsung's Risky Asymmetry [Why&Next]](https://cwcontent.asiae.co.kr/asiaresize/93/2022111609212518207_1668558086.jpg) Home Appliance Woes Mount, Yet 45 Trillion Won ...
Home Appliance Woes Mount, Yet 45 Trillion Won ...
- "A Massive Crash Is Coming in 2026-2027, the Opportunity Is Now"... Advice from ...
- Gyeongbokgung Palace Night Tour Opens on May 13...Traditional Korean Music Perfo...
- The Past of the 'Dumulmeori Body Abandonment' Suspect... Forced a Middle School ...
- 'Maternity Leave for Second Child' Interrupted... 1997-born White House Spokespe...
Professor Cho stated, "With the first step taken toward domestic production of equipment that previously relied entirely on imports, this will help reduce supply instability and enhance continuity of care."
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.







