Samsung Biologics Reports 35% Increase in Q1 Operating Profit to KRW 580.8 Billion (Comprehensive)
Sales Reach 1.2571 Trillion Won
Up 26% Year-on-Year
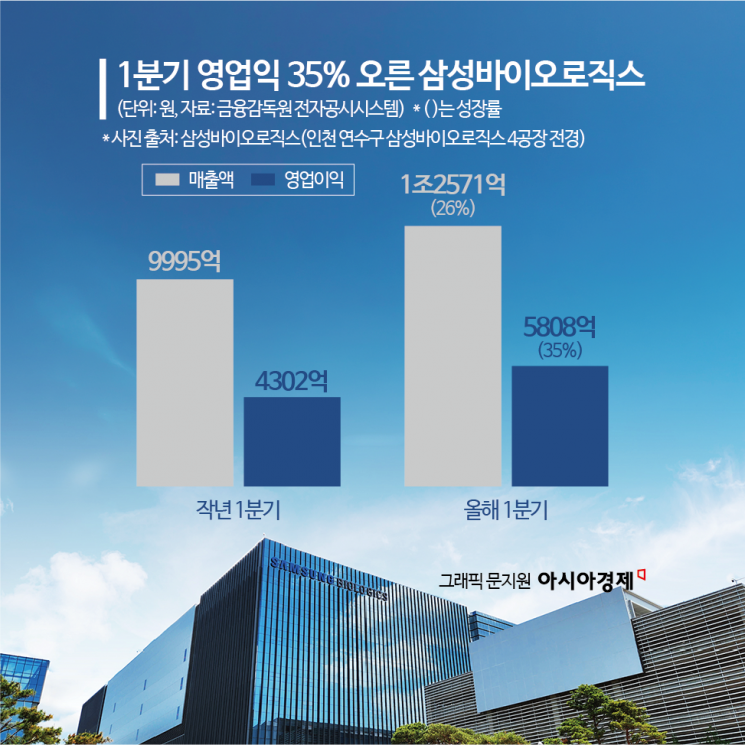
Samsung Biologics posted an operating profit of over 580 billion won in the first quarter of this year. This represents a 35% increase compared to the same period last year.
According to Samsung Biologics’ regulatory filing on April 22, the company reported consolidated sales of 1.2571 trillion won and operating profit of 580.8 billion won for the first quarter of this year. Thanks to full-capacity operation of existing production facilities from Plant 1 to Plant 4, sales rose by 26% and operating profit by 35% year-on-year. The company maintained its annual sales growth guidance at 15–20%, the same level indicated in January. This reflects the continued maximum utilization of existing plants as well as the ramp-up (increase in utilization rate) of Plant 5. As of the end of the first quarter, total assets stood at 11.995 trillion won, equity at 7.9228 trillion won, and liabilities at 4.0722 trillion won. The company maintained a stable financial structure with a debt-to-equity ratio of 51.4% and a borrowing ratio of 11.6%.
845,000-Liter Production Capacity Including Plant 5 and Rockville, U.S.
The overall scale of the business continues to expand through increased production capacity and the establishment of global bases. Plant 5, which began full-scale operation last year with a capacity of 180,000 liters and incorporates optimal operation practices from Plants 1 through 4, is successfully ramping up its utilization rate. As a result, production capacity in Songdo, Incheon now totals 785,000 liters. In particular, at the end of March, the company completed the acquisition of the production facility in Rockville, Maryland, adding an additional 60,000 liters of capacity. The global production capacity now stands at 845,000 liters. By establishing a dual production system connecting Songdo, Korea and Rockville, U.S., Samsung Biologics has secured geographic flexibility and the ability to respond closely to global clients. Revenue contributions from the recently acquired Rockville plant will be announced in future results once reflected in the company’s performance.
Since its founding, the company has secured a total of 112 contract manufacturing (CMO) deals and 169 contract development (CDO) deals, with cumulative orders amounting to USD 21.4 billion (approximately 31.64 trillion won). The company currently counts 17 out of the world’s top 20 pharmaceutical companies as clients. In March, Samsung Biologics signed a contract manufacturing deal worth 279.6 billion won with a European pharmaceutical company, which was disclosed in a regulatory filing. The company continues to actively secure new orders. In January, Samsung Biologics presented its business strategy at the main track of the JP Morgan Healthcare Conference in San Francisco, U.S. The following month, the company participated in DCAT Week 2026, a global pharmaceutical and biotech event held in New York, where it held more than 50 business meetings focused on acquiring new customers.
Expanding Production to ADC, Vectors, and Vaccines Beyond Antibodies
The company is also making concrete progress in diversifying its business portfolio. To respond to the next-generation therapeutics market, Samsung Biologics has invested in a dedicated production facility for antibody-drug conjugates (ADC), moving beyond its traditional antibody-centered portfolio. This facility, which is operated separately from existing antibody production sites due to the handling of cytotoxic drugs, has started commercial production and is playing a key role in portfolio expansion. Additionally, Samsung Biologics has internalized the production of master cell banks (MCB) and vector manufacturing services, establishing an end-to-end service system capable of completing the entire process—from vector construction to investigational new drug (IND) submission—within nine months. The company has also secured flexible vaccine production capabilities based on the Coalition for Epidemic Preparedness Innovations (CEPI) network.
Hot Picks Today
![[Exclusive] "What? I Used It for Fried Eggs and Kimchi Pancakes, but It Wasn't Edible?"... Raw Pork Lard Supplied to Department Stores, Causing Stir](https://cwcontent.asiae.co.kr/asiaresize/93/2026042216151436513_1776842114.png) [Exclusive] "What? I Used It for Fried Eggs and...
[Exclusive] "What? I Used It for Fried Eggs and...
- [Report] "Professionals in Their 30s and 40s With at Least 2 Billion Won in Cash...
- "From 8,000 Won to 730,000 Won: The Toy Everyone Wants but Can't Get"
- "375 Won Per Share" SK hynix to Pay 26.58 Billion Won Cash Dividend
- "Chairman Chey Tae-won's Warning Comes True"... Laptop Prices Already Up 1 Milli...
The company is also strengthening its business foundation in terms of securing future growth engines and sustainable management. In partnership with Eli Lilly, Samsung Biologics has finalized plans to establish a domestic base in Songdo, Incheon for the global pharmaceutical company’s open innovation program, ‘Lilly Gateway Labs (LGL).’ This marks the first time a foreign pharmaceutical company partners with a Korean firm to enter the Korean market, and the base is expected to support the growth and global expansion of promising local biotech companies. In the area of environment, social, and governance (ESG), Samsung Biologics received the highest ‘Platinum’ rating from global evaluation agency EcoVadis. The company has also signed its first ESG contract with a partner and undergone third-party verification for its product carbon footprint calculation system, moving toward not only strengthening its own ESG management capabilities but also enhancing ESG oversight throughout its entire supply chain.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.