"New Drugs Offer False Hope: Cancer and Rare Disease Patients Wait an Average of 3 Years for Insurance Coverage"
Korea Patients Association Analyzes Five-Year Listing Periods
Anti-Cancer Drugs Take 1 Year and 10 Months; Rare Disease Treatments Up to 3 Years and 10 Months
"Introduction of Fast-Track Listing and Responsible Actions from Pharmaceutical Companies Needed"
A recent survey has found that patients suffering from cancer or rare diseases have to wait nearly three years on average to receive newly approved drugs under the national health insurance coverage. Even after treatments are developed, concerns are being raised that patients' lives are being put at risk due to high out-of-pocket costs and complicated administrative procedures.
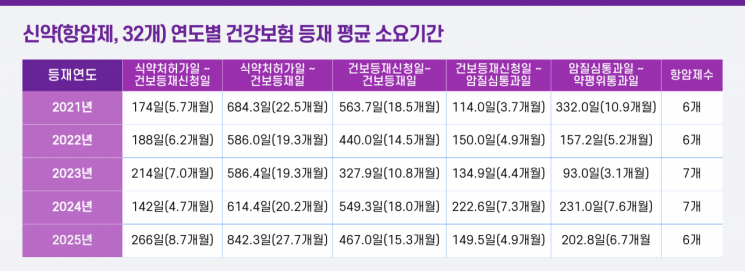
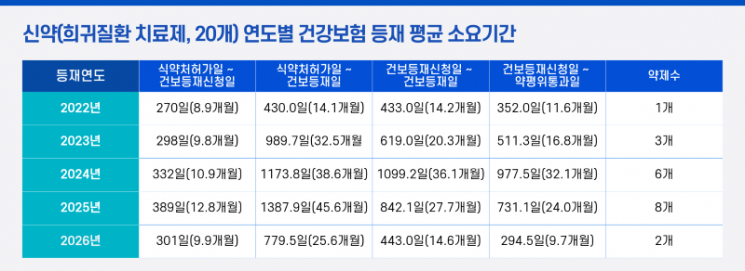
The Korea Patients Association announced on March 10 that it had analyzed the period required for 32 anti-cancer drugs listed under the national health insurance from 2021 to 2025, and 20 rare disease treatments listed from 2022 to 2025, focusing on the time from Ministry of Food and Drug Safety approval to health insurance listing.
According to the analysis, it takes an average of 1 year and 10 months (659 days) for anti-cancer drugs to be covered by health insurance after approval by the Ministry of Food and Drug Safety. For rare disease treatments, the average period from approval to listing was 2 years and 11 months, with some cases delayed as long as 3 years and 10 months.
Currently, the national drug listing process in Korea involves the following steps: approval by the Ministry of Food and Drug Safety, application for listing by the pharmaceutical company, review of reimbursement appropriateness by the Health Insurance Review and Assessment Service, price negotiations with the National Health Insurance Service, and finally, deliberation and announcement by the Health Insurance Policy Deliberation Committee under the Ministry of Health and Welfare. Although the official evaluation period by the Review and Assessment Service is set at 120 to 150 days, in practice, it often takes much longer.
In particular, for anti-cancer drugs, it took an average of 191 days (about six months) from regulatory approval to the pharmaceutical company's application for listing. According to the patient group, this delay occurs because companies postpone applications for strategic negotiation reasons or spend additional time supplementing required documents, considering profitability.
Administrative bottlenecks are also severe. It took about five months to pass the Cancer Disease Review Committee after the application for listing, and a further six to seven months to clear the Drug Reimbursement Assessment Committee. This means that more than a full year is spent at key review stages just to obtain health insurance listing.
The Association pointed out, "For patients with severe illnesses, the time spent waiting for new drugs to be listed is not just a procedural delay, but literally a battle for life." They added, "Despite the existence of treatments, patients are repeatedly forced to give up on treatment or exhaust their assets due to exorbitant out-of-pocket costs." The Association also strongly urged the government to expedite the implementation of the 'Fast-Track Listing and Post-Market Evaluation System for Rare Disease Treatments' currently being promoted. It stated, "Rather than delaying the review due to uncertainty about clinical effectiveness, the paradigm should shift to supplying drugs to patients first and then adjusting or managing the price based on post-market performance."
Hot Picks Today
Ahn Kijong, President of the Korea Patients Association, emphasized, "Pharmaceutical companies must not use patients' desperation as a tool for maximizing profit," and called for "responsible actions such as prompt listing applications after approval and a forward-looking approach to drug pricing."
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.