KAIST Suggests Possibility of Treating Drug Addiction Through Neural Cell Regulation
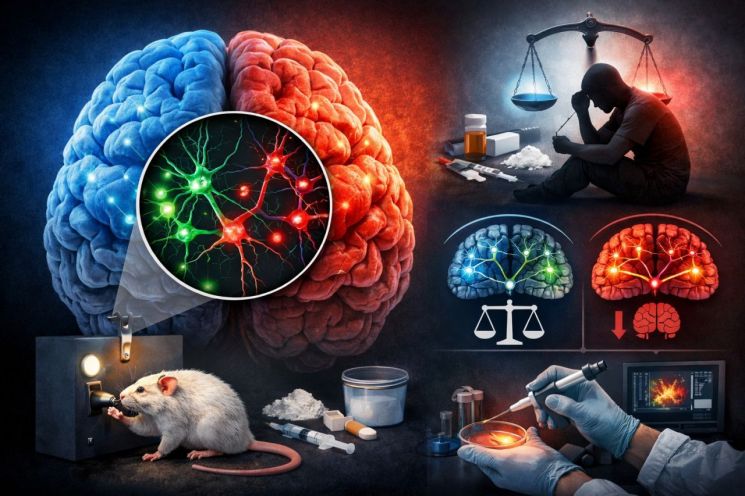
The possibility of treating drug addiction by regulating neural cells has been suggested. Drugs are highly addictive and carry a significant risk of relapse. Until now, the decline in function of the 'prefrontal cortex (PFC)', which controls impulses, has been cited as the cause of this phenomenon. However, recent findings have identified that an imbalance in specific neural cell circuits is the root cause of addiction relapse, opening up the possibility of precision-targeted therapies.
KAIST announced on March 9 that a research team led by Distinguished Professor Sebum Paek from the Department of Brain and Cognitive Sciences at KAIST and Professor Byungkook Lim from the University of California, San Diego (UCSD) has identified the key principle behind how specific inhibitory neural cells in the prefrontal cortex regulate cocaine addiction behavior.
The joint research team focused on 'parvalbumin-positive (PV) inhibitory neural cells', which regulate the balance of neural signals by suppressing the activity of other neurons in the brain. They found that these cells act as a type of 'brake gate' that controls the brain's excitatory signals, and are also a critical factor in determining drug-seeking behavior during withdrawal.
The prefrontal cortex of the brain can perform its 'brake' function to suppress impulses only when excitatory and inhibitory signals are balanced.
To investigate how chronic drug exposure disrupts this balance, the research team conducted experiments in which mice were administered cocaine. This was to track when inhibitory neural cells in the prefrontal cortex are activated and how they send signals to lower brain regions.
The experiments revealed that parvalbumin cells, which account for 60–70% of the inhibitory neurons in the prefrontal cortex, are highly active when the mice attempt to seek cocaine.
However, when the researchers carried out 'extinction training' to teach the mice not to seek the drug, the activity of these cells decreased significantly. This shows that the activity pattern of parvalbumin cells is not permanently fixed by addiction but can be readjusted through extinction processes.
The joint team also conducted experiments in which they artificially regulated neural activity to suppress parvalbumin cell activity. As a result, the cocaine-seeking behavior of the mice decreased dramatically. Conversely, when these cells were activated, drug-seeking behavior persisted even after the extinction process.
This effect was not observed with ordinary rewards such as sugar water, but was specific to drug addiction behavior, indicating that parvalbumin cells are selectively involved in regulating addictive behavior.

(From left) Dr. Minju Jung, Department of Biological Sciences, UCSD; Professor Byungkuk Lim, Department of Biological Sciences, UCSD; Distinguished Professor Sebum Paik, Department of Brain and Cognitive Sciences, KAIST. KAIST
View original imageThe researchers also identified the specific brain circuit through which the regulatory action of parvalbumin cells occurs. They found that signals originating from the prefrontal cortex are transmitted to the reward circuit of the ventral tegmental area (VTA), a core brain region associated with reward, making this pathway a key channel for regulating addictive behavior and deciding whether or not to seek drugs again.
At this point, parvalbumin neurons regulate the flow of this signal and affect dopamine signaling, serving as a kind of 'regulatory switch' that determines whether addictive behavior is maintained or suppressed, according to the joint research team.
In other words, relapse is not due to a decline in the overall function of the prefrontal cortex, but rather determined by how parvalbumin neurons regulate the neural pathways between the prefrontal cortex and the reward circuit.
Distinguished Professor Paek stated, "This research is significant in that it confirms drug addiction is a circuit imbalance problem caused by the collapse of regulatory balance between specific neural cells (parvalbumin cells) and sub-neural circuits. The discovery that parvalbumin cells act as a 'gate' for addictive behavior will provide important clues for developing precision-targeted therapy strategies for drug addiction in the future."
Hot Picks Today
 "Pay for the Postpartum Care Center with My Car...
"Pay for the Postpartum Care Center with My Car...
- "Skipping Diamond Rings... Gen Z in the US Opts for Finger Tattoos"
- "Forged IDs Easily Bypass Convenience Stores, Bars, and Motels... Delinquency En...
- Next Year’s Lottery Sales Expected to Reach Record KRW 8.4 Trillion, Proceeds t...
- Back After 3 Years and 5 Months with a 'Shark Nose Surgery'... Hyundai Unveils T...
Meanwhile, this study was led by Dr. Minjoo Jung at UCSD as the first author, with Professor Byungkook Lim at UCSD and Distinguished Professor Sebum Paek at KAIST serving as co-corresponding authors. The results (paper) were published online in the neuroscience journal 'Neuron' on February 26, 2026.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.