"Bimetallic Atomic Pair Catalyst Converts CO₂ to CO... Demonstrates High-Temperature Stability and Mass Production Potential [Reading Science]"
100% CO Selectivity Achieved at 300-600°C, Near-Equilibrium Conversion
Gram-Scale Synthesis Demonstrates Potential for Process Application
A domestic research team has overcome the stability limitations of high-temperature catalysts for converting carbon dioxide (CO₂) into carbon monoxide (CO), an industrial feedstock, by designing a "bimetallic atomic structure." The team addressed the problem of catalyst metal agglomeration and performance degradation at high temperatures through atomic-level structural design, and demonstrated gram-scale synthesis, presenting the potential for process application.
On March 8, it was announced that Dr. Hyuntak Kim, Senior Researcher at the Korea Research Institute of Chemical Technology, together with Professor Youngjin Kim of Kyungpook National University, Professor Geunsik Lee of Ulsan National Institute of Science and Technology (UNIST), and Professor Sangjun Kim of Chungnam National University, developed a bimetallic atomic catalyst with metal atoms designed in paired form. This catalyst enables stable conversion of CO₂ to CO even in high-temperature thermochemical reactions.
Examples of Carbon Dioxide (CO2) and Carbon Monoxide (CO) Conversion and Utilization in Household Products. Provided by the Research Team
View original imageThe results of this research were published in the international journal "Nature Communications." Researcher Kyungmin Kim of Korea Research Institute of Chemical Technology and Researcher Jinhong Moon of UNIST participated as first authors, while Dr. Hyuntak Kim, Professor Youngjin Kim, Professor Geunsik Lee, and Professor Sangjun Kim contributed as corresponding authors.
Limitations of High-Temperature Catalysts in CO₂ Conversion Processes
The technology for converting CO₂ into CO is considered a key process in the production of synthetic fuels and chemical products. CO is a major component of syngas and serves as the starting material in the production processes of methanol, synthetic fuels, plastics, and chemical feedstocks.
In particular, when combined with green hydrogen (H₂) based on renewable energy, it becomes a core step in the "Power-to-X" process, which converts electricity into fuel and chemicals.
However, CO₂ is a chemically very stable molecule. The conversion reaction typically requires high temperatures above 500-600°C, and during the reaction, the phenomenon of "sintering," where catalyst metal particles cluster together, easily leads to performance degradation.
Previously, metal nanoparticle catalysts such as nickel (Ni), copper (Cu), and platinum (Pt) have been used, but they suffered from performance drop and by-product formation during long-term high-temperature operation. Although there have been studies on single-atom catalysts (SAC) that disperse metals in single-atom form, issues remained with metal atoms migrating or agglomerating in reducing environments.
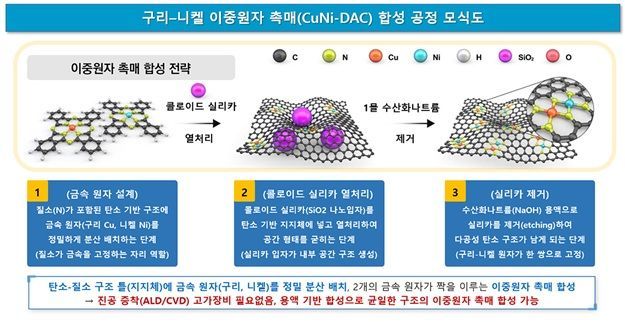
Schematic diagram of the bimetallic catalyst synthesis process. Provided by the research team
View original imageSecuring Both Selectivity and Stability Through Atomic Pair Design
The research team developed a bimetallic atomic catalyst (Cu?Ni DAC), in which copper and nickel are precisely arranged in adjacent atomic pairs, rather than in bulk form.
They designed a structure where the two metal atoms are fixed within a nitrogen-doped carbon framework, forming an N₂Cu-N₂-NiN₂ configuration. This structure promotes CO₂ activation, rapidly separates the produced CO, and suppresses the formation of methane (CH₄).
Experimental results showed that this catalyst achieved nearly 100% selectivity for CO within the 300-600°C range, with almost no formation of by-products such as methane.
Furthermore, it maintained stable performance for more than 100 hours even under harsh conditions involving repeated temperature changes.
The reverse water-gas shift (RWGS) reaction, which converts CO₂ into CO, has thermodynamic limits, but this catalyst achieved a conversion rate of 64% under experimental conditions, which is close to the theoretical equilibrium conversion rate of 66%.

Photo of the joint research team. From the center left clockwise: Dr. Hyuntak Kim, Korea Institute of Science and Technology, corresponding author; Professor Youngjin Kim, Kyungpook National University, corresponding author; Professor Geunsik Lee, UNIST, corresponding author; Professor Sangjun Kim, Chungnam National University, corresponding author; Researcher Kyungmin Kim, Korea Institute of Science and Technology, first author; Researcher Jinhong Moon, UNIST, first author. Provided by Korea Institute of Science and Technology
View original imageGram-Scale Synthesis Achieved...Potential for Process Application
Another notable feature of this research is the potential for mass production.
Most previous single-atom catalyst studies have been limited to milligram-scale laboratory synthesis. The research team developed a synthesis strategy using solution mixing, drying, and heat treatment, instead of expensive equipment such as vacuum deposition (ALD, CVD).
With this method, they successfully achieved repeated production of bimetallic atomic catalysts on a 13-15 gram scale using the same process. They explained that this suggests potential for application in existing thermocatalytic processes, such as industrial fixed-bed reactors.
Dr. Hyuntak Kim, Senior Researcher at the Korea Research Institute of Chemical Technology, said, "By precisely designing the Cu-Ni bimetallic atomic structure, we confirmed that CO₂ can be selectively converted into CO even under high-temperature conditions, while maintaining the atomic dispersion structure during repeated operation."
Hot Picks Today
 "Could I Also Receive 370 Billion Won?"... No Limit on 'Stock Manipulation Whistleblower Rewards' Starting the 26th
"Could I Also Receive 370 Billion Won?"... No Limit on 'Stock Manipulation Whistleblower Rewards' Starting the 26th
- Samsung Electronics Labor-Management Reach Agreement, General Strike Postponed... "Deficit-Business Unit Allocation Deferred for One Year"
- "From a 70 Million Won Loss to a 350 Million Won Profit with Samsung and SK hynix"... 'Stock Jackpot' Grandfather Gains Attention
- "Stocks Are Not Taxed, but Annual Crypto Gains Over 2.5 Million Won to Be Taxed Next Year... Investors Push Back"
- "Who Is Visiting Japan These Days?" The Once-Crowded Tourist Spots Empty Out... What's Happening?
Lee Youngguk, President of the Korea Research Institute of Chemical Technology, stated, "This study overcame the stability limitations of atomic catalysts and demonstrated the feasibility of mass production. We expect it to contribute to strengthening the competitiveness of carbon resource technology."
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.







