Pharmaceutical Approvals Decline for Fifth Year... Biosimilars Reach Record High
MFDS Approves 1,197 Pharmaceutical Products Last Year
Steady Increase in Orphan Drug Approvals
Active Development of AI-Based Domestic SaMD
Although the number of domestic pharmaceutical approvals and notifications decreased last year, approvals for biosimilars (equivalent biological products) reached an all-time high.
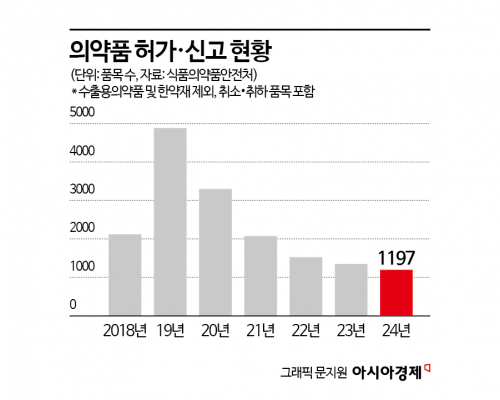
According to the Ministry of Food and Drug Safety's report on the status of approvals, certifications, and notifications for pharmaceuticals, quasi-drugs, and medical devices released on April 29, a total of 1,197 pharmaceutical products were approved or notified in 2024. This represents an 11.3% decrease (152 products) from the 1,349 products in 2023, marking a continued decline for the fifth consecutive year.
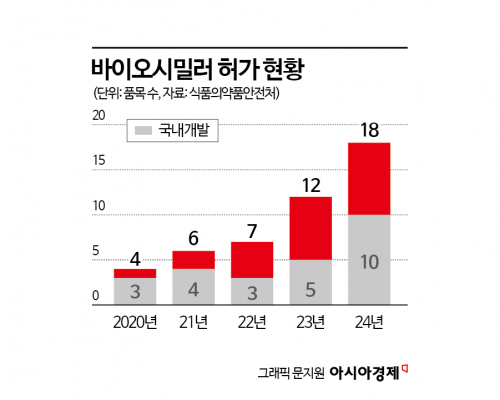
Among pharmaceuticals, 18 biosimilar products (comprising 10 active ingredients) were approved, an increase of 6 products from the previous year. This is the highest number of biosimilar approvals since the first product was approved in 2012.
Of these, more than half?13 products (7 active ingredients)?were developed domestically. As a result, out of the total 72 biosimilar products (35 active ingredients) approved up to last year, approximately 72%, or 52 products (24 active ingredients), were domestically developed. The Ministry of Food and Drug Safety expects domestic companies to remain active in biosimilar development, as many original products by the initial developers are approaching patent expiration.
The number of approvals and notifications for generic drugs and similar products increased by 43 from the previous year, reaching 845 products. Since July 2021, when the number of times identical clinical trial data could be used for different products was limited to three, the annual number has remained at a similar level for the past three years. The Ministry interprets this as evidence that the system has stabilized.
The number of generic drug approvals and notifications dropped from 2,813 products in 2020 to 1,614 products in 2021, and has remained in the 800s since 2022.
A total of 39 orphan drugs (26 active ingredients) were approved last year, an increase of 2 products from the previous year. Among these, antineoplastic agents, including treatments for acute myeloid leukemia, accounted for the largest share at 16 products.
Last year, circulatory system drugs containing Ginkgo biloba leaf dry extract as the main ingredient were the most frequently approved, with 161 products. This was followed by antipyretic, analgesic, and anti-inflammatory drugs (146 products), antidiabetic agents (127 products), other vitamin preparations (99 products), and antineoplastic agents (39 products).
There were a total of 659 quasi-drug products approved or notified. Domestically manufactured products accounted for 567 cases, making up 86% of all approvals and notifications in this category. By product group, sanitary pads had the highest proportion at 41.1% (271 products), followed by toothpastes at 19.3% and adhesive bandages at 10.5%.
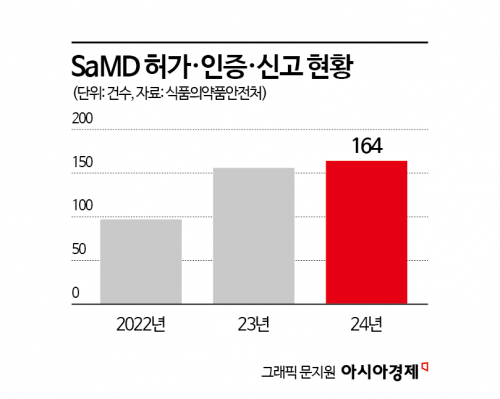
A total of 7,116 medical device products were approved, certified, or notified. Among these, approvals, certifications, and notifications for standalone medical device software (SaMD) have steadily increased over the past three years, reaching 164 cases last year alone. Notably, as the performance of artificial intelligence (AI)-based SaMD has improved, a number of first-in-Korea approvals have been granted in various clinical fields. Of the 41 innovative medical devices approved up to last year, 33 products (80.5%) were SaMD.
In addition, as interest in skin aesthetics and plastic surgery continues to rise both domestically and internationally, medical devices related to skin?such as those for tissue repair and wrinkle improvement?are being consistently approved. Furthermore, due to the aging population and the resulting increase in the prevalence of tooth loss and related diseases, products primarily used by older adults, such as "dental implant superstructures" (135 cases, 3rd most frequent) and "dental implant surgical instruments" (98 cases, 6th most frequent), were also included among the top approved and certified items in 2024.
Hot Picks Today
![[Breaking] Trump: "Bombing Resumes if Iran Deal is Rejected... Sanctions Lifted if Agreement is Reached"](https://cwcontent.asiae.co.kr/asiaresize/93/2026050422131350385_1777900394.png) [Breaking] Trump: "Bombing Resumes if Iran Deal...
[Breaking] Trump: "Bombing Resumes if Iran Deal...
- "Stock Set to Double: This Company Smiles Every Time a Data Center Is Built [Cli...
- "Continuous Groundwater Pumping Causes Mexico City to Sink 24cm Annually... 'Gia...
- “She Shouted, ‘The Rope Isn’t Tied!’... Chinese Woman Falls from 168m Cliff ...
- "Prime Minister in Underwear?"... Italy's Meloni Posts Herself to Warn of Deepfa...
A Ministry of Food and Drug Safety official stated, "We will continue to actively provide useful information related to medical product approvals so that consumers can stay informed, and we will do our utmost to ensure that more safe and high-quality medical products are developed."
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.