Development of Low-Temperature, Low-Pressure Ammonia Synthesis Catalyst with 7 Times Higher Productivity
Professor Min-Ki Choi's Team Develops a High-Performance Catalyst for Ammonia Synthesis
Achieves Over Seven Times Higher Productivity Under Mild Conditions
Enables Decentralized, Eco-Friendly Ammonia Production for the Hydrogen Economy
Researchers at the Korea Advanced Institute of Science and Technology (KAIST) have developed a high-performance catalyst capable of synthesizing ammonia without energy loss even at very low temperatures and pressures.
On the 11th, Professor Min-Ki Choi's research team from KAIST's Department of Bio and Chemical Engineering announced that they have developed an innovative catalytic system that can drastically reduce energy consumption and carbon dioxide emissions while significantly increasing ammonia productivity.
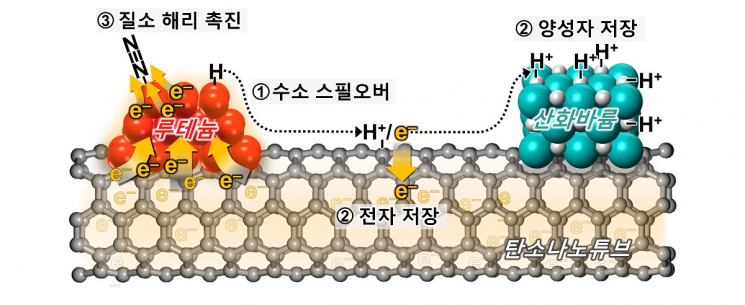
Schematic diagram illustrating the mechanism of enhanced catalytic activity of ruthenium catalyst by barium oxide co-catalyst. Provided by KAIST
View original imageHydrogen production using renewable energy is a key technology for eco-friendly energy generation. However, hydrogen produced in this way is difficult to store and transport, so it is stored in the form of ammonia (NH3), which has no carbon emissions and is easy to liquefy.
Ammonia is produced using iron (Fe)-based catalysts through the Haber-Bosch process, a technology over 100 years old. This process involves high temperature and high pressure (500℃, 100 atm), resulting in enormous energy consumption, large amounts of carbon dioxide emissions, and high distribution costs.
Therefore, recently, an eco-friendly process synthesizing ammonia at low temperature and low pressure (300℃, 10 atm) using green hydrogen produced by water electrolysis has attracted attention. However, this process requires the development of catalysts that can secure high ammonia productivity at low temperature and low pressure. Currently, ammonia productivity through this process is low, and overcoming this is a key challenge.
The research team developed a novel catalyst that acts like a "chemical capacitor" by introducing ruthenium (Ru) catalysts and strongly basic barium oxide (BaO) particles onto a highly conductive carbon surface.

From the left, Baek Ye-jun, PhD candidate in the Department of Bio and Chemical Engineering at KAIST, and Professor Choi Min-gi. Provided by KAIST
View original imageDuring the ammonia synthesis reaction, hydrogen molecules (H2) dissociate into hydrogen atoms (H) on the ruthenium catalyst, and these hydrogen atoms further dissociate into proton (H+) and electron (e-) pairs. The acidic protons are stored in the strongly basic barium oxide, while the remaining electrons are separately stored in ruthenium and carbon.
Through this unique chemical capacitor phenomenon, the electron-rich ruthenium catalyst promotes the dissociation of nitrogen (N2) molecules, which is the key step in ammonia synthesis, thereby dramatically enhancing catalytic activity.
In particular, this study discovered that by controlling the nanostructure of carbon, the electron density of ruthenium can be maximized, further enhancing catalytic activity. Under mild conditions of 300℃ and 10 atm, this catalyst exhibited ammonia synthesis performance more than seven times higher than the best existing catalysts.
Professor Min-Ki Choi stated, "This study confirmed that efficient ammonia synthesis is possible under low temperature and low pressure conditions using high-performance catalysts. This will enable decentralized small-scale ammonia production, moving away from the conventional large-scale factory-centered production, and allow more flexible ammonia production and utilization suitable for an eco-friendly hydrogen economy system."
Hot Picks Today
 "If It's Uncomfortable, They Cut Ties": Three O...
"If It's Uncomfortable, They Cut Ties": Three O...
Professor Choi served as the corresponding author, and PhD student Yejun Baek was the first author of the study. The research results were published on the 24th of last month in the prestigious international journal in the field of catalytic chemistry, Nature Catalysis. (Paper title: Electron and proton storage on separate Ru and BaO domains mediated by conductive low-work-function carbon to accelerate ammonia synthesis)
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.







