Celltrion's 'Uplima' Accelerates Entry into US Market... "Covering 50% of Market"
Leading the 3.5 Trillion Sales with Jimpentra
Breaking Through Two of the Three Major PBMs as the 'Gateway to the US Market'
Accelerating Entry with Strategic Changes Including Dual Pricing
Celltrion's autoimmune disease treatments, Uplyma and Jimptetra, which are at the forefront of its goal to achieve annual sales of 3.5 trillion KRW this year, are accelerating their penetration into the U.S. market.
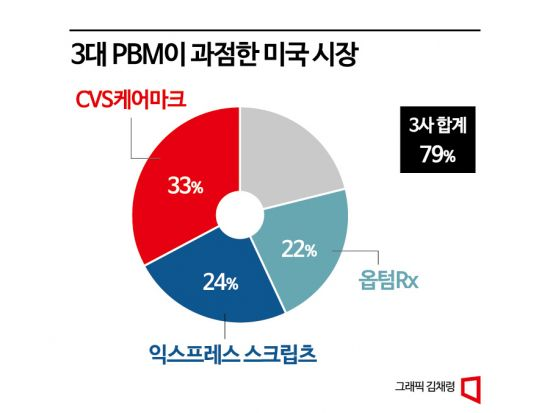
On the 12th, Celltrion announced that Uplyma, developed as a biosimilar to Humira, recently signed a formulary listing contract with one of the three major U.S. pharmacy benefit managers (PBMs). In the U.S. market, if Uplyma is not listed on the PBM's formulary associated with the patient's insurer, doctors find it difficult to prescribe Uplyma to patients. This is because the PBM guarantees reimbursement only for drugs listed on the formulary; otherwise, patients must bear the full cost of the medication. The U.S. PBM market is currently dominated by three major PBMs?CVS Caremark, Express Scripts, and OptumRx?which collectively control 80% of the market.
However, since its launch last year, Uplyma has only succeeded in being listed on OptumRx among these PBMs. As a result, it has faced challenges in market penetration. As of February, Uplyma's market share in the U.S. was around 0.1%. Compared to the original Humira's 96% market share and other biosimilars, this is a low figure. But with this second major PBM contract, the company has succeeded in increasing its coverage in the U.S. market to 50% at once. The company plans to secure a listing contract with the remaining PBM within the year to significantly expand coverage.
This contract is particularly notable because Celltrion has significantly changed its sales strategy for Uplyma. Instead of the high wholesale acquisition cost (WAC) strategy used until last year, it introduced a 'dual pricing' policy that sells products at both high and low wholesale prices. This is due to the characteristics of the U.S. market, where rebates are allowed, unlike in Korea. The rebate amount paid to PBMs is calculated as a percentage of the wholesale price. The higher the wholesale price, the greater the PBM's profit. Naturally, PBMs prefer products with higher wholesale prices. Until now, Celltrion had adopted a single pricing strategy with a wholesale price of $6,577, which is only a 5% discount compared to Humira's $6,922 per month. In contrast, companies like Amgen, Sandoz, and Boehringer Ingelheim, which have launched high wholesale price products, have implemented a dual pricing strategy by simultaneously selling low wholesale price products discounted by up to 87%.
Starting this year, Celltrion plans to make dual pricing its core strategy. The company explained, "The most important factor when signing PBM contracts is profitability," adding, "We have established a pricing structure that maximizes profitability by supplying high wholesale price products to the public insurance market, where rebate rates are low, and low wholesale price products to the private insurance market, where rebate levels are high." The recent PBM contract, covering both public and private insurance, was made for the low wholesale price product, and Celltrion is expected to focus more on the private insurance market.
This is also a tailored strategy considering the U.S. Inflation Reduction Act (IRA), which will be implemented from next year. Through the IRA, the U.S. government is actively pushing for drug price reductions, requiring insurers to pay 60% of the excess cost for drugs priced over $2,000 annually. As a result, from next year, market preference is expected to shift toward lower wholesale price products rather than higher-priced ones. Celltrion believes that "in anticipation of regulatory changes, we quickly listed the low wholesale price product with fewer rebates on major PBMs with high market shares to secure a more advantageous position in market penetration."
To achieve its sales target of 3.5 trillion KRW this year, Uplyma needs to generate 500 billion KRW and Jimptetra 1 trillion KRW globally. Celltrion has also created conditions to sell the two drugs together as a package in the U.S. Both drugs are used to treat autoimmune diseases such as inflammatory bowel disease (IBD). Since doctors will have the option to prescribe the drug more suitable for the patient between the two, it is expected to contribute to market expansion. Furthermore, with the launch of CT-P43, a biosimilar of another autoimmune disease treatment, Stelara, scheduled for March next year, a stronger therapeutic portfolio is expected to be established.
Hot Picks Today
!["Stock Set to Double: This Company Smiles Every Time a Data Center Is Built [Click e-Stock]"](https://cwcontent.asiae.co.kr/asiaresize/93/2026050416112750184_1777878687.jpg) "Stock Set to Double: This Company Smiles Every...
"Stock Set to Double: This Company Smiles Every...
- "Is Yours Just Gathering Dust at Home? Millennials & Gen Z Rediscover Digicams O...
- "Continuous Groundwater Pumping Causes Mexico City to Sink 24cm Annually... 'Gia...
- "I Take Full Responsibility"... Seongjae Ahn Issues Direct Apology for 'Wine Swi...
- “She Shouted, ‘The Rope Isn’t Tied!’... Chinese Woman Falls from 168m Cliff ...
Additionally, from this contract onward, Celltrion plans to expand Uplyma's target market beyond inflammatory bowel disease to include rheumatoid arthritis (RA), another key indication of Humira. This is expected to enable entry into the overall U.S. market, where Humira's sales reached $14.44 billion (approximately 20 trillion KRW) last year.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
![Celltrion's Humira biosimilar Uplyma <span>[Photo by Celltrion]</span>](https://cphoto.asiae.co.kr/listimglink/1/2023091809195679039_1694996395.jpg)