UNIST Develops Method to Improve Hydrogen Fuel Cell Efficiency Using Ion Materials
Researchers at UNIST have developed a method to revolutionarily improve the efficiency of hydrogen fuel cells, which are gaining attention as an eco-friendly next-generation energy source.
Professor Myungsoo Na's team in the Department of Chemistry developed a solid electrolyte material using Metal-Organic Frameworks (MOF). Through this, they innovatively enhanced the conductivity of hydrogen ions within the solid electrolyte used in hydrogen fuel cells.
The research team was the first to introduce guest molecules with low acidity among the guest molecules that provide hydrogen ions and serve as mediators. They also developed a new methodology to increase the number of guest molecules inside the MOF pores, thereby improving hydrogen ion conductivity.
Hydrogen fuel cells are high-efficiency and eco-friendly power generation devices that directly convert chemical energy generated from the chemical reaction of hydrogen and oxygen into electrical energy.
The currently mainly used Proton-Exchange Membrane Fuel Cell uses Nafion, which has thermal, mechanical, and chemical stability, as the electrolyte to achieve high hydrogen ion conductivity.
However, it has the disadvantage of a low operating temperature threshold and an unclear mechanism, limiting performance improvement.
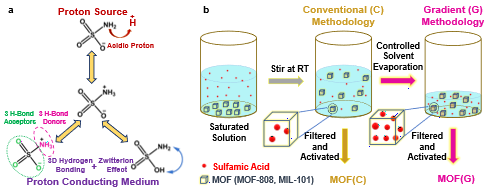
Characteristics of sulfamic acid and explanation of superprotonic ion conductivity a (left), a new methodology for introducing sulfamic acid into MOF pores b.
View original imageThe research team focused on MOF as an alternative. MOF is a material formed by the combination of metals and organic compounds to create a porous structure, and it has recently attracted attention as a material for fuel cells due to its excellent chemical and thermal stability.
MOFs have pores of various sizes formed during synthesis, and guest molecules that enhance hydrogen ion conductivity can be introduced into these pores, making them useful for developing materials with high hydrogen ion conductivity.
The research team introduced sulfamic acid, an amphoteric ion substance with low acidity and both positive and negative charges, as a guest molecule into two types of MOFs (MOF-808, MIL-101). The guest molecule sulfamic acid can form various types of hydrogen bonds, acting as an excellent mediator for hydrogen ion transfer.
The team developed, for the first time, a material with high hydrogen ion conductivity (above 10^-1 S cm^-1) by increasing the amount of sulfamic acid inside the MOF pores. They also confirmed that the hydrogen ion conductivity is maintained over a long period, securing high durability.
Professor Myungsoo Na of the Department of Chemistry said, “This research presented a new paradigm that removes the limitation of having to use MOFs stable to strong acidic substances in developing highly conductive hydrogen ion materials,” adding, “The study using the amphoteric ion sulfamic acid provides new opportunities for application in various fields.”
Dr. Amitosh Sharma and Dr. Jaewoong Lim of UNIST’s Department of Chemistry participated as co-first authors in this research, which was conducted under the Mid-Career Researcher Support Program led by the Ministry of Science and ICT and the National Research Foundation of Korea.
Hot Picks Today
!["Stock Set to Double: This Company Smiles Every Time a Data Center Is Built [Click e-Stock]"](https://cwcontent.asiae.co.kr/asiaresize/93/2026050416112750184_1777878687.jpg) "Stock Set to Double: This Company Smiles Every...
"Stock Set to Double: This Company Smiles Every...
- "Is Yours Just Gathering Dust at Home? Millennials & Gen Z Rediscover Digicams O...
- "Continuous Groundwater Pumping Causes Mexico City to Sink 24cm Annually... 'Gia...
- "I Take Full Responsibility"... Seongjae Ahn Issues Direct Apology for 'Wine Swi...
- “She Shouted, ‘The Rope Isn’t Tied!’... Chinese Woman Falls from 168m Cliff ...
Additionally, on May 26, the research was published online in Angewandte Chemie International Edition, a world-renowned journal in the field of chemistry, and was recognized for its excellence by being selected as the Back cover paper on June 20.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.







