DeepCure Receives MFDS Clinical IDE Approval for Resistant Hypertension Treatment Device
DeepCure, a company specializing in laparoscopic renal denervation (RDN) devices, is entering clinical trials for a resistant hypertension treatment device.
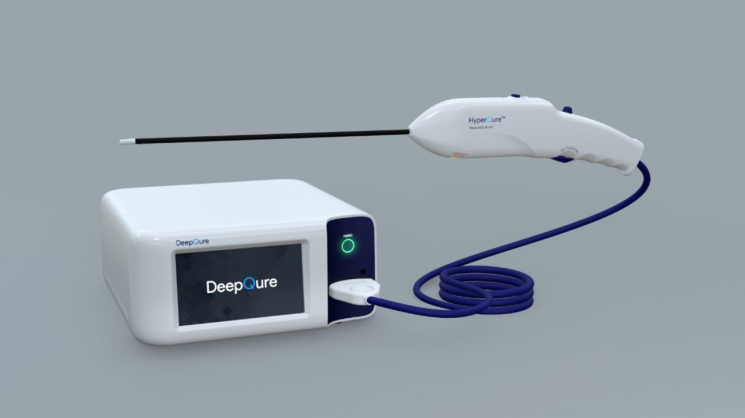
On the 17th, DeepCure announced that it received approval from the Ministry of Food and Drug Safety for the clinical trial application (IDE) of the laparoscopic RDN medical device 'HyperQure™.'
The clinical trial will evaluate the safety and initial efficacy of laparoscopic renal denervation using HyperQure in patients with resistant hypertension who are taking three or more types of antihypertensive medications. The trial will be conducted prospectively, as a single-arm, open-label study at Yonsei University College of Medicine Severance Hospital, Catholic University Seoul St. Mary's Hospital, Seoul National University Hospital, Hwasun Chonnam National University Hospital, and Hanyang University Hospital.
HyperQure is a resistant hypertension treatment device that uses a laparoscopic approach to block the sympathetic nerves by applying a radiofrequency (RF) electrode device in contact with the renal artery. Unlike the catheter method that emits radiofrequency energy inside the renal artery, HyperQure wraps around the blood vessel externally and delivers radiofrequency energy to block the sympathetic nerves. This overcomes the limitations of the existing catheter method, such as the risk of vascular intimal damage and incomplete nerve blockade.
A company representative stated, "This is the first clinical trial applying a laparoscopic RDN medical device to humans in Korea. Starting with domestic clinical trials, we will thoroughly prepare for global trials to prove the safety and efficacy of 'HyperQure' and obtain approval as the world's first resistant hypertension treatment device."
Hot Picks Today
!["Stock Set to Double: This Company Smiles Every Time a Data Center Is Built [Click e-Stock]"](https://cwcontent.asiae.co.kr/asiaresize/93/2026050416112750184_1777878687.jpg) "Stock Set to Double: This Company Smiles Every...
"Stock Set to Double: This Company Smiles Every...
- "Is Yours Just Gathering Dust at Home? Millennials & Gen Z Rediscover Digicams O...
- "Continuous Groundwater Pumping Causes Mexico City to Sink 24cm Annually... 'Gia...
- "I Take Full Responsibility"... Seongjae Ahn Issues Direct Apology for 'Wine Swi...
- “She Shouted, ‘The Rope Isn’t Tied!’... Chinese Woman Falls from 168m Cliff ...
DeepCure is a bio-venture company developing the world's first laparoscopic RDN medical device. In the first half of this year, it plans to apply for clinical approval from the U.S. Food and Drug Administration. To ensure successful domestic and international clinical progress, the company is actively recruiting global experts with extensive experience and expertise in urology and cardiology as members of its Scientific Advisory Board (SAB).
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.