RSV Fatal to Infants but Without Vaccine... Big Pharma's All-Out Development Effort
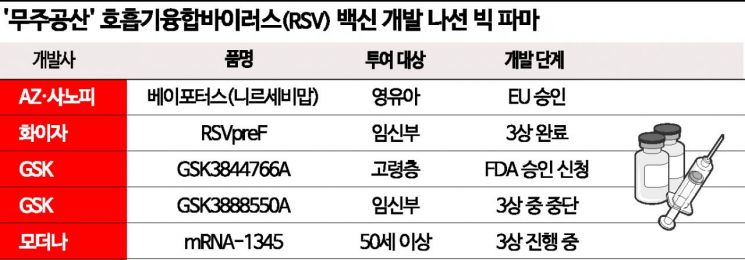
AZ·Sanofi Jointly Developed 'Vepotus'
EU Approved... RSV and Lower Respiratory Infection Reduced by 75%
Pfizer Completes Phase 3 for Pregnant Women
GSK Applies for FDA Approval for Elderly
Moderna Develops mRNA-Based 'COVID-19 and Trivalent Flu Vaccine'
SK Bioscience Also Pursues mRNA-Based Development
[Asia Economy Reporter Chunhee Lee] Respiratory syncytial virus (RSV) is spreading globally, but no preventive vaccine has been developed yet. With leading global big pharma companies racing to develop RSV vaccines, the launch of the first vaccine is now within sight.
According to industry sources on the 7th, the pharmaceutical companies most advanced in RSV vaccine development are AstraZeneca (AZ) and Sanofi. The single-dose RSV preventive antibody treatment for infants, Beyfortus (generic name: nirsevimab), jointly developed by AZ and Sanofi, recently received approval from the European Union (EU) Executive Commission (EC), becoming the first general-use RSV preventive drug. Although AZ and AbbVie received approval for the preventive antibody treatment Synagis (generic name: palivizumab) in 1998, it was limited only to infants with heart and lung diseases.
AZ and Sanofi stated that Beyfortus reduced the incidence of lower respiratory tract infections caused by RSV by 74.5% over 150 days in Phase 3 clinical trials. Neutralizing antibody levels were about 50 times higher than baseline on day 151 after administration.
Vaccines administered to pregnant women during the fetal stage rather than after birth are also being researched. Pfizer recently completed Phase 3 clinical trials of RSVpreF (PF-06928316) and released the results. It showed an 81.8% prevention rate for severe lower respiratory tract infections and a 57.1% prevention rate for overall lower respiratory tract infections in infants within 90 days of birth. GlaxoSmithKline (GSK) was also developing a vaccine for pregnant women, GSK3888550A, but halted clinical trials due to safety concerns raised during the process.
Instead, GSK is expediting approval for its vaccine targeting the elderly, GSK3844766A. Phase 3 clinical trials confirmed overall efficacy against RSV lower respiratory tract infections in adults aged 60 and over. The company has submitted a Biologics License Application (BLA) to the U.S. Food and Drug Administration (FDA). Designated for priority review, the approval decision is expected by May next year. GSK has also applied for approval from the European Medicines Agency (EMA) and Japan’s Ministry of Health, Labour and Welfare.
Moderna has embarked on developing an RSV vaccine using messenger RNA (mRNA). Under the development name mRNA-1345, it is conducting global Phase 3 clinical trials involving 34,000 participants aged 60 and above. Moderna is also conducting trials for those aged 50 and over and is developing a trivalent vaccine, mRNA-1230, which aims to prevent RSV, COVID-19, and influenza simultaneously.
Hot Picks Today
!["Not Everyone Can Afford This: Inside the World of the True Top 0.1% [Luxury World]"](https://cwcontent.asiae.co.kr/asiaresize/93/2026051507261063923_1778797570.png) "Not Everyone Can Afford This: Inside the World of the True Top 0.1% [Luxury World]"
"Not Everyone Can Afford This: Inside the World of the True Top 0.1% [Luxury World]"
- While All Eyes Were on Samsung and Hynix, This Company Surged 50% to New Highs in Four Days [Weekend Money]
- "Now Our Salaries Are 10 Million Won a Month" Record High... Semiconductor Boom Drives Performance Bonuses at Major Electronic Component Firms
- "Sold Out Everywhere" The Surprising Story of the 'Purple Gold' Philippine Yam That Has Captivated the World [Delicious Stories]
- Experts Already Watching Closely..."Target Price Set at 970,000 Won" Only Upward Momentum Remains [Weekend Money]
In South Korea, SK Bioscience is preparing to develop an RSV vaccine. Although still in the basic research stage, the company recently announced plans to develop vaccines against RSV and other diseases using an mRNA vaccine platform in collaboration with the Coalition for Epidemic Preparedness Innovations (CEPI).
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
![A baby is receiving a vaccination. <br>[Photo by Yonhap News]](https://cphoto.asiae.co.kr/listimglink/1/2022102608185782331_1666739937.jpg)