Who Is the Second Domestic COVID-19 Vaccine?
SK Bioscience's 'GBP510' Achieves First Domestic Phase 3 Clinical Success
Green Light for Follow-up Development
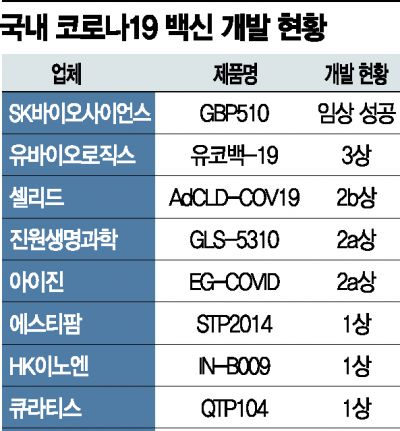
Competition Among 8 Companies Including UbioLogics, Cellid,
Jinwon Life Sciences, and Quratis in Vaccine Development
Ubio's 'Ucovac-19'
Plans to Complete Phase 3 Clinical Trial by Q3 This Year
Most Promising as Second Domestic Vaccine
[Asia Economy Reporter Lee Chun-hee] With SK Bioscience’s ‘GBP510’ becoming the first domestically developed COVID-19 vaccine to successfully complete Phase 3 clinical trials, a green light has been turned on for the previously sluggish development of follow-up domestic vaccines. Attention is also focused on which company will succeed in developing the second domestic vaccine.
Sequential Clinical Trials by Companies Including YuBio
According to the Ministry of Food and Drug Safety on the 27th, a total of eight companies are pursuing COVID-19 vaccine development. Following SK Bioscience, YuBiologics, Cellid, GeneOne Life Science, Quratis, HK inno.N, iGen, and ST Pharm are all aiming to develop the second domestic COVID-19 vaccine.
The most likely next contender is YuBiologics’ ‘Yukobac-19’. Except for GBP510, it is the only domestically developed vaccine to have successfully entered Phase 3 clinical trials. In January, it received approval from the Ministry of Food and Drug Safety for a multinational Phase 3 clinical trial involving 4,000 participants in Korea, the Philippines, and Bangladesh. The company plans to complete the Phase 3 trials overseas by the third quarter of this year. Cellid also aimed to simultaneously apply for Phase 2b and 3 clinical trials for ‘AdCLD-COV19’, but after consultations with the Ministry of Food and Drug Safety, decided to proceed with Phase 2b first and submit Phase 3 separately later.
Vaccine development based on messenger RNA (mRNA) technology, which is attracting attention as a next-generation vaccine, is also underway. ST Pharm (STP2014), iGen (EG-COVID), and Quratis (QTP104) are pursuing mRNA COVID-19 vaccine development. In particular, ST Pharm has formed the ‘K-mRNA Consortium’ together with Hanmi Pharmaceutical, GC Green Cross, and Dong-A ST. Additionally, a structure called the ‘mRNA Bio Venture Consortium’ has been formed, where iGen, Quratis, and GeneOne Life Science handle technology development, and Boryung Biopharma undertakes contract manufacturing (CMO), leading to competition between the two consortia for market dominance.
Some companies have shifted to vaccines dedicated to booster shots. GeneOne Life Science has downsized the existing Phase 2a trial of ‘GLS-5310’ and plans to proceed with Phase 2b and 3 trials as a booster-only vaccine based on the safety and efficacy confirmed through this. Park Young-geun, CEO of GeneOne Life Science, explained, “There have been reports of limitations in the preventive efficacy of existing COVID-19 vaccines upon re-administration, and it is also expected that annual vaccinations may be necessary.” GeneOne Life Science is also conducting Phase 2 trials of the therapeutic agent ‘GLS-1027’. SK Bioscience is also conducting clinical trials for booster and heterologous vaccinations of GBP510.
GBP510 Success Acts as a Catalyst for Follow-up Vaccine Development
The success of GBP510 is expected to have a positive impact on the development of other vaccines. Human clinical trials are conducted by comparing an experimental group administered the target drug with a control group given a placebo such as saline. However, in the pandemic situation, administering a placebo is difficult, so current domestic Phase 3 trials use existing approved vaccines as comparator vaccines in the control group, comparing clinical data to prove non-inferiority. GBP510 also used the AstraZeneca (AZ) vaccine as the comparator vaccine in its Phase 3 trials.
On the 25th, when the 4th COVID-19 booster vaccination for those aged 60 and over who made advance reservations began, a citizen received the vaccine at Bumin Hospital in Gangseo-gu, Seoul. April 25, 2022 Photo by Joint Press Corps (portrait consent obtained)
View original imageHowever, it has been practically difficult to use vaccines developed overseas as comparator vaccines, causing many domestic companies to face difficulties in conducting Phase 3 trials. Industry consensus is that SK Bioscience would have struggled to secure a comparator vaccine without its prior contract manufacturing (CMO) of the AZ vaccine and government support. YuBiologics has succeeded in securing the AZ vaccine as a comparator overseas, making Phase 3 administration visible, but has yet to secure a comparator vaccine domestically. However, if GBP510 is actively used as a domestic comparator vaccine, these difficulties are expected to be somewhat alleviated.
Hot Picks Today
!["Stock Set to Double: This Company Smiles Every Time a Data Center Is Built [Click e-Stock]"](https://cwcontent.asiae.co.kr/asiaresize/93/2026050416112750184_1777878687.jpg) "Stock Set to Double: This Company Smiles Every...
"Stock Set to Double: This Company Smiles Every...
- "Continuous Groundwater Extraction Causes Mexico City of 22 Million to Sink by 2...
- “Did They Bet Too Early?” Losses Snowball for ‘Geopverse Ants’ as KOSPI Soar...
- Axios: US and Iran Near One-Page Agreement... Awaiting Iran's Response (Update)
- "Prime Minister in Underwear?"... Italy's Meloni Posts Herself to Warn of Deepfa...
Meanwhile, some companies have abandoned vaccine development. Genexine withdrew the clinical trials of its vaccine candidate ‘GX-19N’, which had been approved for Phase 2 and 3 trials in Indonesia last month. This decision was based on the assessment that the business viability of development was low considering the COVID-19 epidemic situation and vaccine market supply and demand.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.