Novocellbio Launches Full-Scale Clinical Trials of NK Cell Therapy in Mexico
[Asia Economy Reporter Chunhee Lee] Novocell Bio announced on the 12th that it has completed the NK cell culture experiment of Novocell Bio's NK cell therapy (NOVO-NK) conducted at Monterrey Zambrano Hellion Hospital to obtain final clinical trial approval from Mexico's Texsalud and the Mexican Food and Drug Administration, and will commence full-scale clinical trials.
The two companies previously completed a cell therapy manufacturing facility within Zambrano Hellion Hospital last month. Sara Gonzalez, spokesperson for Texsalud, stated that they will start full-scale clinical trials and patient treatment in both countries in connection with Novocell Bio's biopharmaceutical manufacturing facility in Korea.
Hot Picks Today
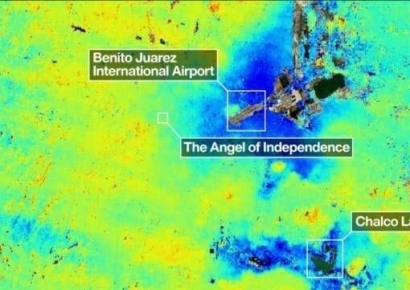 "Continuous Groundwater Extraction Causes Mexic...
"Continuous Groundwater Extraction Causes Mexic...
- "Stock Set to Double: This Company Smiles Every Time a Data Center Is Built [Cli...
- "Is Yours Just Gathering Dust at Home? Millennials & Gen Z Rediscover Digicams O...
- "Going to Seongsu-dong?" Japanese Girl Group Faces Taxi Refusal in Seoul
- "Prime Minister in Underwear?"... Italy's Meloni Posts Herself to Warn of Deepfa...
First, clinical trials for COVID-19 will begin in the first half of this year, followed by additional clinical trials for breast cancer in the second half. Subsequently, they plan to expand the indications.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.








