The Most Approved Vaccines Worldwide: AZ, Pfizer, Sputnik in Order
[Asia Economy Reporter Lee Seon-ae] The most widely authorized COVID-19 vaccine worldwide is the AstraZeneca (AZ) vaccine. It is followed by the Pfizer vaccine and the Russian Sputnik V vaccine.
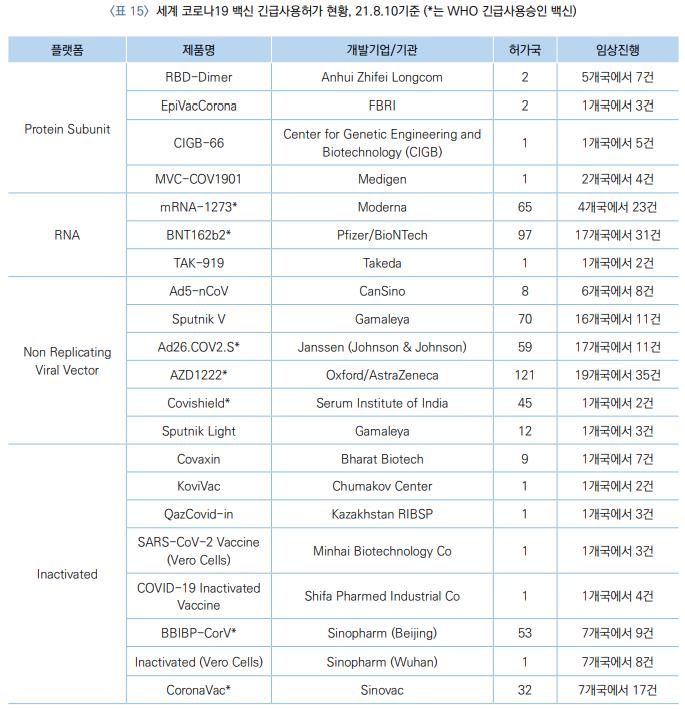
According to the Ministry of Food and Drug Safety (MFDS) on the 22nd, as of the 10th of this month, there are 21 COVID-19 vaccines that have received emergency use authorization globally. Among them, 7 vaccines have received emergency use approval from the World Health Organization (WHO).
The AstraZeneca vaccine (AZD1222) has been approved in 121 countries worldwide, making it the COVID-19 vaccine used in the most countries. It also has the highest number of clinical trials, with 35 trials conducted in 19 countries.
The vaccine developed by Pfizer and BioNTech (BNT162b2) and the Sputnik V vaccine developed by the Gamaleya Research Institute under the Russian Ministry of Health were approved in 97 and 70 countries respectively, ranking second and third.
The AstraZeneca and Pfizer vaccines were officially approved in South Korea in February and March of this year, respectively. The Sputnik V vaccine is being contract-manufactured by Huons [243070] in South Korea, which has applied for preliminary review of product approval with the MFDS, but the formal review process has not yet started.
The Moderna vaccine (mRNA-1273) and the Janssen (Johnson & Johnson) vaccine (Ad26.COV2.S), which are authorized and administered domestically, have been approved in 65 and 59 countries respectively.
Additionally, the vaccine developed by Chinese pharmaceutical company Sinopharm (BBIBP-CorV) is approved in 53 countries, the AstraZeneca vaccine (Covishield) contract-produced by Indian pharmaceutical company Serum Institute of India (SII) in 45 countries, and the vaccine developed by Chinese pharmaceutical company Sinovac (CoronaVac) in 32 countries.
Furthermore, among global vaccine research and development, COVID-19 preventive vaccines account for 111 cases, representing 22%, making it the most active area. Seasonal flu vaccines account for 38 cases (7%), cancer vaccines 33 cases (6%), and human papillomavirus (HPV) vaccines 17 cases (3%), following behind.
By company, the UK’s GSK leads with 19 research and development projects, followed by France’s Sanofi, China’s CanSino Biologics, South Korea’s Genexine and its US affiliate NeoImmuneTech, US-based Epivax, Moderna, and Inovio.
Hot Picks Today
 In the End, Even Wafer Pods Were Removed Over Fears of a Strike... Samsung Electronics Enters 'Emergency Mode'
In the End, Even Wafer Pods Were Removed Over Fears of a Strike... Samsung Electronics Enters 'Emergency Mode'
- No Bacteria Detected in Arisu After 24 Hours of Repeated Drinking from a Tumbler
- "Available Only in Korea": Pokémon Card Prices Surge 2,532% Due to Rarity, Becoming Investment Assets
- “He Did What Even Family Couldn’t”… Teacher Supports Student Who Lost Father for 7 Years
- "Addicted to Uncovering Hidden Value Investing Opportunities"... Korean Stock Fever Sweeps the US
This statistic was published in the '2021 First Half Vaccine Industry Latest Trends Report' jointly issued by the MFDS, the Vaccine Safety Technology Support Center, and the Korea BioPharmaceutical Association.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
![[Image source=Yonhap News]](https://cphoto.asiae.co.kr/listimglink/1/2021082210243986291_1629595479.jpg)