Making Money While Saving the Planet? ... The Transformation of Carbon Dioxide, the Main Culprit of Climate Disaster
UNIST Develops Ultra-Low Voltage Electrochemical System for Converting Carbon Dioxide to Formic Acid
Productivity Up, Power Consumption Down Through Paired Reaction Replacement
Published in Angewandte Chemie
A new production system has been developed that converts carbon dioxide, the main culprit of global warming, into formic acid, a high-value-added chemical. This technology transforms carbon dioxide from a "carbon disaster" into an "environmental hero."
The system reduces power consumption to one-fourth of previous levels while nearly tripling production output.
On November 12, a team led by Professor Seungho Cho from the Department of Materials Science and Engineering, and Professors Youngguk Kwon and Jaesung Lee from the Department of Energy and Chemical Engineering at UNIST announced that they have developed an ultra-low voltage electrochemical system that converts carbon dioxide into formic acid.

Research team, (from left) Professor Seungho Cho, Professor Youngguk Kwon, Professor Jaesung Lee, Researcher Hyoseok Kim, Dr. Wonsik Jang, Dr. Jinho Lee, Researcher Hojeong Lee. Provided by UNIST
View original imageBy applying electricity to carbon dioxide to convert it into formic acid, it is possible to both reduce greenhouse gases and produce a high-value-added substance. However, a significant amount of electricity is wasted in this process.
This is due to the inefficiency of the "oxygen evolution reaction," which is the paired reaction. In reaction systems that convert carbon dioxide to formic acid, oxygen is generated as a result of the paired reaction, and producing this oxygen consumes 70-90% of the total power, causing the system operating voltage to rise up to 2V.
The research team developed a system that replaces the problematic oxygen evolution paired reaction with a formaldehyde oxidation reaction (FOR). This system can produce formic acid at both electrodes with high efficiency (96.1% at the cathode, 82.1% at the anode) even at a low voltage of 0.5V. When the system operating voltage is reduced to one-fourth, power consumption decreases accordingly.
Furthermore, the total production rate of formic acid reached 0.39 mmol/cm²·h, nearly three times higher than that of existing systems. Using the formaldehyde oxidation reaction as the paired reaction enables the production of formic acid, rather than oxygen, at the paired reaction electrode.
The team was able to develop this system by creating a new copper-silver composite catalyst essential for the formaldehyde oxidation reaction. Conventional catalysts used for this reaction suffer a rapid decline in activity.
The formaldehyde oxidation reaction using the new catalyst can also be applied to the development of eco-friendly, self-powered systems for producing ammonia, hydrogen peroxide, and hydrogen without electricity. In practice, the researchers combined the formaldehyde oxidation reaction with the nitrate reduction reaction, oxygen reduction reaction, and hydrogen evolution reaction, respectively, and succeeded in producing ammonia, hydrogen peroxide, and hydrogen without electricity or pollutant emissions.
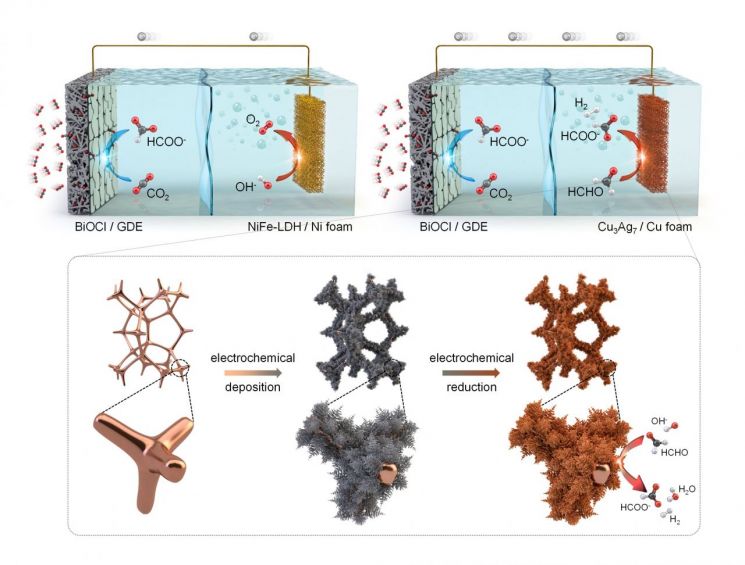
New system converting carbon dioxide into formic acid (top right) and the structure of the developed catalyst (bottom).
View original imageProfessor Seungho Cho stated, "This technology eliminates the greatest inefficiency in carbon dioxide conversion and maximizes the use of limited electrical energy. In addition to carbon dioxide conversion, it can be applied to various eco-friendly chemical process technologies, contributing to solving environmental and resource circulation issues."
Hyoseok Kim, Wonsik Jang, and Jinho Lee from the Department of Materials Science and Engineering, as well as Hojeong Lee from the Department of Energy and Chemical Engineering, participated as first authors in this research.
The research results were published online on October 1 in the renowned international journal in the field of comprehensive chemistry, "Angewandte Chemie International Edition," and have been selected as a cover paper, with formal publication forthcoming.
Hot Picks Today
This research was supported by the UNIST InnoCORE program and the National Research Foundation of Korea.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.







