First in Korea to Receive US FDA De Novo Approval... UNIST Startup Resense Medical's OcuCool, an Ocular Anesthesia Medical Device
Rapid Precision Cooling Technology Proven Safe and Effective ... Alternative to Conventional Chemical Anesthesia
Innovation Based on UNIST Research, Licens Medical Develops World's First New Medical Technology
Licens Medical, a company founded by Professor Kim Gunho of the Department of Mechanical Engineering at UNIST (President Park Jongrae), has recently received De Novo approval from the U.S. Food and Drug Administration (FDA) for its ophthalmic contact cooling anesthesia device, OcuCool®.
This is the first time a domestic medical device has received De Novo approval.
The FDA De Novo process is an approval procedure applied to new medical devices with no existing equivalent, rigorously verifying the safety and efficacy of innovative technologies. OcuCool attracted attention as an alternative to traditional chemical anesthesia by passing this stringent process.
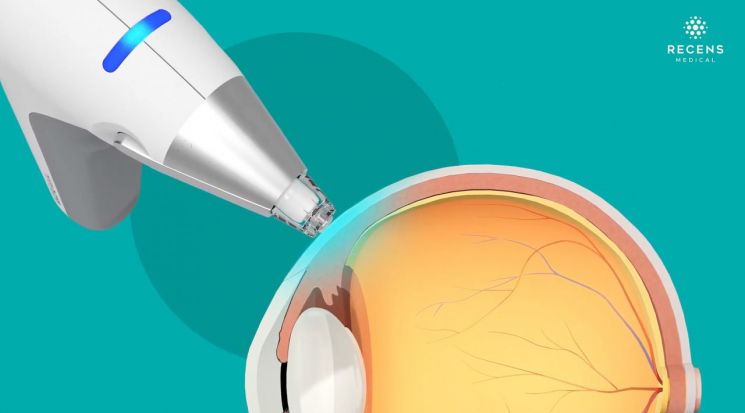
OcuCool utilizes rapid precision cooling technology that safely blocks nerve signal transmission. When applied to the treatment area, anesthesia takes effect in just 10 seconds. Compared to conventional chemical anesthetics, which take about 5 to 10 minutes, this greatly reduces procedure time.
The total procedure time has been reduced by about tenfold, from the conventional 10-15 minutes to just 1-2 minutes. By minimizing exposure to chemical substances, side effects such as redness, stinging, and dryness are also reduced. For the treatment of blinding diseases like macular degeneration and diabetic retinopathy, it is safer and more effective than existing injection procedures (IVT: Intravitreal Injection).
The safety and efficacy of OcuCool were demonstrated through phase 1 to 3 clinical trials conducted at 10 ophthalmology hospitals in the U.S. from 2018 to 2022. Eighty percent of patients participating in the trials preferred OcuCool over conventional anesthetics, as they directly experienced shorter procedure times and fewer side effects.
Charles Wykoff, a specialist at Houston Retina Consultants, explained, "Cooling anesthesia produces rapid effects through physical action," adding, "Patients expressed great satisfaction with the shorter procedure times and faster recovery."
With the aging population and increasing number of diabetes patients, the market for intravitreal injection (IVT) procedures using OcuCool is growing. According to market research firm QY Research, the global IVT market is expected to reach approximately $22 billion (about 29.6 trillion KRW) by 2027.
Kim Gunho, CEO of Licens Medical, stated, "The FDA De Novo approval for OcuCool is an achievement based on research results from UNIST," adding, "We are pleased to provide faster and more comfortable procedures for patients with vision loss. As a company leading innovative technology rather than following others, we aim to showcase the excellence of Korean medical devices to the world and contribute to patients and medical professionals with more innovative products."
Licens Medical was established in 2016 as a faculty startup from UNIST and developed the world’s first rapid precision cooling medical device. Currently, its dermatology cooling device, TargetCool®, is sold in 29 countries, and the company is preparing to enter the hair loss treatment and animal/home-use medical device markets. It also aims for a KOSDAQ technical listing next year.
Through this achievement, UNIST has confirmed that university research outcomes can lead to real innovation in the industry. The university plans to further strengthen support for startups and technology commercialization in the future.
Hot Picks Today
 "If It's Uncomfortable, They Cut Ties": Three O...
"If It's Uncomfortable, They Cut Ties": Three O...
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.