Cancer-targeting 'Nuclear Missile'... Next-Generation Radiolabeled Targeted Cancer Drug Development Race
Radioactive Drug Delivery for Cancer
Shorter Half-Life than ADC
Overcoming Resistance with Minimal Drawbacks
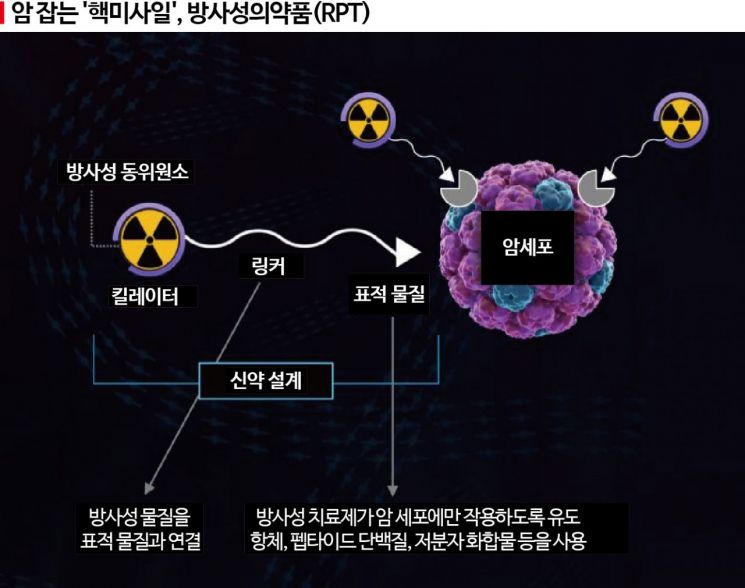
The pace of advancement in targeted cancer therapies that selectively attack cancer cells is accelerating. While antibody-drug conjugates (ADCs), which deliver 'poison' exclusively to cancer cells, are leading the global pharmaceutical and biotech industries, competition is now heating up in radiopharmaceutical therapy (RPT), which delivers 'radioactive drugs' to cancer.
ADCs are called 'cruise missiles' that precisely target cancer by combining antibodies that bind to cancer cells with toxic drugs that kill cancer. The breast cancer treatment 'Enhertu' is a representative example. Although the treatment is highly effective, the antibody’s half-life lasts 2 to 3 weeks, which means it can remain in the body for too long, and the toxic drugs may induce resistance, posing limitations.
RPT, which uses radioactive isotopes, overcomes the drawbacks of ADCs and enhances effectiveness. Because RPT utilizes radioactivity, it is often likened to a 'guided nuclear missile.' When injected into cancer patients, it initially detects and targets cancer cells precisely, similar to ADCs. Upon reaching the cancer cells, the radioactive isotopes emit radiation to attack the cancer cells. With a half-life of only 7 to 11 days and almost no resistance, it is expected to overcome the limitations of ADCs.
Among global big pharma companies, Novartis leads the RPT market. In 2017 and 2018, Novartis acquired two RPT developers for a total of $5.9 billion (approximately 8 trillion KRW), securing the prostate cancer treatment 'Pluvicto' and the neuroendocrine tumor treatment 'Lutathera,' both of which have received approval from the U.S. Food and Drug Administration (FDA). Especially, with Pluvicto’s approval in April 2022, the RPT market officially expanded. Pluvicto reduced patients’ risk of death by 38% and doubled progression-free survival (PFS) to 8.7 months compared to existing treatments.
![Novartis' Radiopharmaceutical Therapy (RPT) for Prostate Cancer 'Pluvicto' <br>[Photo by Novartis]](https://cphoto.asiae.co.kr/listimglink/1/2024012309011538285_1705968075.png)
Novartis' Radiopharmaceutical Therapy (RPT) for Prostate Cancer 'Pluvicto'
[Photo by Novartis]
The global RPT market is expected to grow from $6.5 billion in 2021 at an average annual growth rate of 6%, reaching $11.2 billion (approximately 15 trillion KRW) by 2030. Other big pharma companies such as Bristol Myers Squibb (BMS) and Eli Lilly are also securing related technologies. Last month, BMS acquired RPT developer Rays Bio for $4.1 billion (approximately 5.5 trillion KRW), and on the 17th, invested $50 million (approximately 67 billion KRW) in Retio Therapeutics. Retio holds technology related to the alpha particle radioactive isotope Actinium (Ac)-225. Alpha particles have higher energy than beta particles currently used in external radiation therapy, enabling more effective cancer treatment, but their poor penetration into the human body has prevented their use in conventional radiation therapy. However, by applying RPT to directly inject alpha particles into the patient’s body, cancer treatment becomes possible. Last October, Eli Lilly acquired Point Biopharma for $1.4 billion (approximately 1.876 trillion KRW).
Domestically, SK Biopharm is actively investing in RPT. Through TerraPower, a small modular reactor (SMR) company invested in by SK Inc., it secured rights to Ac-225 in Asia, ensuring stable supply of radioactive isotopes. Lee Dong-hoon, CEO of SK Biopharm, said, “We decided to enter the RPT field by leveraging our ability to procure radioactive isotopes,” adding, “We are reviewing business directions including in-house new drug development.” Additionally, FutureChem is conducting Phase 2 clinical trials of the prostate cancer treatment 'FC705.' Since radioactive isotopes can also be used for diagnostic products depending on their application, 'FC303' for prostate cancer diagnosis is undergoing Phase 3 clinical trials. Other companies such as Dukem Bio and Selbion are also developing RPT.
Hot Picks Today
!["What If the KOSPI Plummets?... Record Funds Flow into 'Target Maturity Funds' [Investment Trends]"](https://cwcontent.asiae.co.kr/asiaresize/93/2026043008433546440_1777506215.jpg) "What If the KOSPI Plummets?... Record Funds Fl...
"What If the KOSPI Plummets?... Record Funds Fl...
- "Parents Deposited 10 Million Won for Me"... Securities Accounts Surge 272% Amid...
- Is a Bigger Home Really Necessary When a 59㎡ Unit Offers the Same Layout for 30...
- "If It's Uncomfortable, They Cut Ties": Three Out of Five U.S. Gen Z Have Broken...
- Did Her Wealthy Husband Quash the Investigation? Yang Jungwon Appears Before Pol...
However, challenges remain, such as the short half-life of radioactive isotopes. While a short half-life reduces the risk of side effects by limiting the time the drug stays in the body, it can pose obstacles to commercialization. Because the drug must be administered to patients within a few days of production, if perfect 'just-in-time production' is not achieved, the drug’s efficacy may decrease.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.