'Drugs to Prevent Aging' Enter Their First Target Year... First Patient Dosed in the US [New Path for Bio, Anti-Aging] ①
by Park Jeongyeon
Published 13 May.2026 10:00(KST)
Updated 13 May.2026 11:21(KST)
First Clinical Trial for Epigenetic Reprogramming Begins
Consensus Reached on 14 Aging Biomarkers for Clinical Trials
① "The Fight Against Aging" Becomes a Global Next-Generation Industry
② The Anti-Aging Drug That Even Big Pharma Could Not Crack—A Window of Opportunity for Korea
③ Why Did Samsung's Anti-Aging Research, Once Ahead of Google, Fall 13 Years Behind?
④ K-BigHeart Dreams of Becoming the Korean IMEC
2026 is considered the first year in which aging itself has become the direct target of new drug development. In January, Life Biosciences in the United States received Investigational New Drug (IND) approval from the U.S. Food and Drug Administration (FDA) for its epigenetic reprogramming therapy "ER-100" and began dosing the first patient in March. This marks the first time in history that a drug designed to reverse aging itself has been administered to a human. What was once the private domain of Silicon Valley billionaires has now officially become part of the R&D pipeline of the global pharmaceutical industry. Aging is no longer just an academic pursuit but is being redefined as a new drug development market attracting massive capital.
According to Longevity Technology, a UK-based longevity industry platform, global anti-aging biotech funding in the first quarter of 2026 reached 3.74 billion dollars (approximately 5.5 trillion won), a sharp 56% increase year-on-year. The number of deals also rose from 43 to 49. Annual investment is projected to reach 9 billion dollars (about 13.265 trillion won). As regulatory doors are opening for clinical entry, investment capital is also pouring in at a rapid pace.
Anti-aging has already entered the mainstream R&D domain of big pharma. According to Aging Biotech Info, a non-profit research group, the cumulative disclosed deal size for big pharma in the anti-aging sector has surpassed 11 billion dollars (around 16.331 trillion won). The major deal leading this influx of funds is the collaboration agreement between Eli Lilly and Insilico Medicine. In March, the two companies signed a new drug development partnership worth up to 2.75 billion dollars (about 4 trillion won). Lilly paid an upfront payment of 115 million dollars (about 170.7 billion won) and secured global exclusive rights to Insilico’s preclinical-stage oral drug candidates, which were discovered using an AI platform based on aging biology. Novartis has launched its Aging & Regenerative (DARe) Medicine division and is collaborating with the anti-aging biotech company BioAge in a partnership worth up to 550 million dollars (about 816.4 billion won).
There is also emerging consensus on how to clinically measure aging. Last year, the international journal "Journal of Gerontology" published 14 aging biomarkers identified by international aging researchers through a Delphi consensus process. These include molecular markers such as IGF-1, GDF-15, hsCRP, and IL-6; functional indicators such as muscle mass, grip strength, and gait speed; and epigenetic clocks like DunedinPACE. In March of this year, a Berlin research team reported in "Biomarker Research" that among the 14 biomarkers, epigenetic aging pace best predicted mortality. This means that the primary outline of a standard measurement tool for clinical trials has taken shape.
Korea Joins the Anti-Aging Front—Both Government and Industry Move Together
In Korea, R&D targeting aging directly as a new drug indication has begun simultaneously in both the public and private sectors. In its 2026 Healthcare R&D Implementation Plan announced in December last year, the Ministry of Health and Welfare launched a new project for "anti-aging and rejuvenation regenerative medicine translational clinical research." This is a six-year translational clinical program aiming to reach IND approval for Phase 1 clinical trials. It is the first project to allocate budget for R&D on cell and gene therapies that directly target aging, not just as a consequence of disease.
Hanmi Pharm, a traditional pharmaceutical company and R&D powerhouse, has presented anti-aging as its next growth engine after obesity therapeutics. The company is focusing on the latest research trends showing that GLP-1-based obesity drugs may also delay aging by reducing inflammation and neuroinflammation pathways. This is similar to how Lilly seeks its next engine in aging biology after GLP-1. Hanmi is incorporating anti-aging efficacy evaluation items into clinical protocols for incretin-based drugs and is developing its own omics-based biological age measurement tools.
Among biotechs, Medipost—the world’s first to commercialize an allogeneic stem cell therapy—has found a clue for its new anti-aging business in its existing cord blood pipeline. After establishing an anti-aging research team in 2023, the company began verifying the anti-aging effects of cord blood plasma. According to a study recently published in the international journal "Rejuvenation Research," cord blood plasma significantly reduced the expression of genes related to aging. The company plans to expand into new businesses targeting skin regeneration and anti-aging using components derived from cord blood.
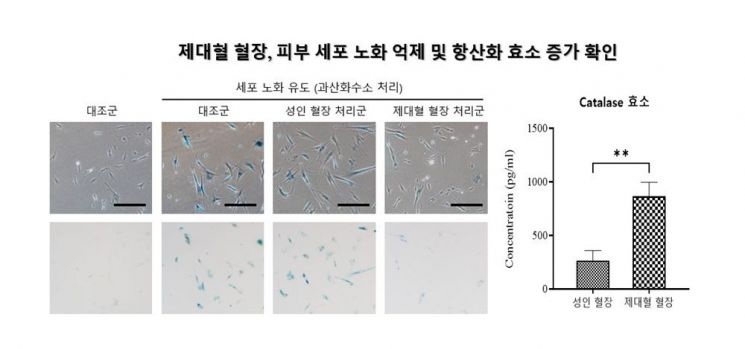
Research results confirming the inhibition of skin cell aging and increase of antioxidant enzymes through umbilical cord blood plasma. Medipost
View original image"The Next Two Years: Data, Not Hype, Will Decide the Winners"
Experts predict that the next one to two years will determine the fate of the anti-aging sector. This is because key clinical results, the outcome of early investment recoveries, and big pharma’s next portfolio decisions are all expected to converge during this period. Clinical data will come first. The initial Phase 1 ER-100 signals from Life Biosciences are expected at the end of this year or early next year. IND entry for the Lilly–Insilico drug candidate, as well as Phase 1 and 2 data from BioAge and Rubedo, are also expected to be released in the same period. The clinical validity of the entire aging-targeted drug category will be put to the test for the first time.
The capital market is also expected to demand returns. The 5.5 trillion won inflow in the first quarter will put the first-generation biotechs’ series maturities and big pharma partnership milestones under the microscope simultaneously. Unity Biotechnology, a first-generation senolytics company, raised 200 million dollars through Series C funding but shut down its business after clinical failure. Big pharma’s decision-making clocks are also ticking faster. Cumulative blockbuster revenue that will expire by the early 2030s exceeds 300 billion dollars. The GLP-1 market is projected to reach 150 billion dollars by 2030, but after that, aging biology is essentially the only remaining mega category candidate.
An industry source said, "Aging-targeted new drugs have moved beyond the stage of possibility verification and are now at the stage of establishing the category," and added, "Clinical data from the next one to two years will determine the direction of big pharma’s growth engines after GLP-1."

!['Drugs to Prevent Aging' Enter Their First Target Year... First Patient Dosed in the US [New Path for Bio, Anti-Aging] ①](https://cphoto.asiae.co.kr/listimglink/1/2026051209285058820_1778545730.jpg)