Samil Pharmaceutical Swings to Loss, Faces Double Blow from CDMO Delays and Key Product Drop
by Lee Sungmin
Published 06 Feb.2026 08:59(KST)
Global GMP Certification Delays for Vietnam CMO Plant
"Full-Scale Commercial Production After Obtaining KGMP in the First Half of the Year"
A warning light has come on for Samil Pharmaceutical's profitability. Its key product, Gliptide tablets, which used to generate around 10 billion won in annual sales, has effectively entered a phase of being dropped from prescriptions. As its Vietnam ophthalmic solution contract development and manufacturing (CDMO) business has also failed to begin commercial production due to delayed certifications, concerns are mounting over the company's ability to defend its short-term performance.
According to the industry on the 6th, the Ministry of Food and Drug Safety distributed an information letter to doctors, pharmacists, and patients the previous day, recommending discontinuation of use and switching to alternative medicines for sucralfate glycotide formulations based on the results of a re-evaluation of drug safety and efficacy. While no safety issues were found, regulators concluded that efficacy in treating gastric and duodenal ulcers, as well as gastritis and duodenitis, had not been demonstrated.
Samil Pharmaceutical is the only company in Korea that has Gliptide tablets, a sucralfate glycotide formulation. Gliptide tablets account for about 5% of Samil Pharmaceutical's total sales, with annual revenue of approximately 10 billion won. Along with Livact (a nutritional supplement for patients with liver cirrhosis), Foributin (a gastrointestinal motility modulator), and Monoprost (an ophthalmic solution for glaucoma), it is one of the company's flagship products.
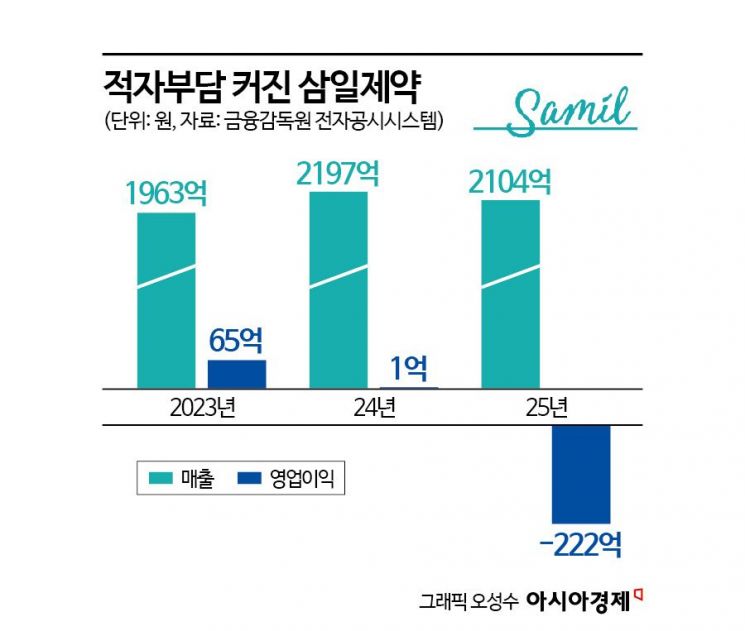
The problem is that this negative development has coincided with a period of weak earnings. On a consolidated basis last year, Samil Pharmaceutical posted sales of 210.4 billion won and an operating loss of 22.2 billion won, swinging into the red. Sales fell 4.2% from a year earlier. The company cited declining sales of major products, increased research and development expenses, and higher selling, general, and administrative expenses related to preparing for commercial production and obtaining GMP certification at its Vietnamese subsidiary as factors behind the deteriorating results.
Another burden is that the Vietnam ophthalmic solution CDMO business, into which Samil Pharmaceutical has invested around 130 billion won, has yet to translate into visible revenue. After establishing a subsidiary in Vietnam in 2018, the company completed a CDMO plant in 2022 capable of producing about 300 million units of ophthalmic solutions annually. However, there is an assessment that the transition to commercial production is being delayed as certification procedures for full-scale operation have been pushed back.
In the global contract manufacturing organization (CMO) and CDMO business, securing Good Manufacturing Practice (GMP) certification is a prerequisite for receiving orders from clients and starting commercial production. Samil Pharmaceutical has not yet completed all of the key certification procedures. It obtained Vietnam GMP in 2024, but the Korean KGMP certification it had targeted afterward is being delayed. It has also not yet secured the U.S. Food and Drug Administration (FDA) cGMP certification or the European Union EU-GMP certification. The company expects that once it obtains KGMP certification this year, commercial production will gradually ramp up.
Ultimately, with sales growth of existing products stagnating, many observers believe that Samil Pharmaceutical's profitability recovery hinges on whether CDMO commercial production following GMP certification this year can actually translate into revenue. A Samil Pharmaceutical official said, "In the global CMO and CDMO pharmaceutical business, high quality and cost competitiveness are key," adding, "We plan to secure cost competitiveness based on Vietnam's low labor costs, maintain quality competitiveness through our know-how in ophthalmic drug production, and nurture this as a mid- to long-term growth engine."