Kyung Hee University Professor Do Kyung Kim's Team Achieves Breakthrough in Cancer Research with Copper-Iron Nanocomposites
by Choi Youngchan
Published 23 Sep.2025 17:18(KST)
Kyung Hee University announced on the 23rd that a research team led by Professor Do Kyung Kim from the College of Medicine has introduced a new cancer treatment strategy that can overcome the conventional 'apoptosis' resistance of cancer cells.

From the left, Professor Kim Dokyung of Kyung Hee University College of Medicine, Research Professor Cha Byungseok. Kyung Hee University
View original imageThe research team systematically analyzed that a nanocomposite combining copper (Cu) and iron (Fe) metal ions with ligands can act exclusively on cancer cells to induce two novel forms of cell death: 'cuproptosis' (copper-dependent cell death) and 'ferroptosis' (iron-dependent cell death).
The research findings were published in the international academic journal 'Coordination Chemistry Reviews' (Impact Factor: 23.5, JCR top 1%) under the title 'Beyond Apoptosis: Navigating Cancer Therapy with Cu/Fe-Ligand Nano-complexes through Cuproptosis and Ferroptosis.'
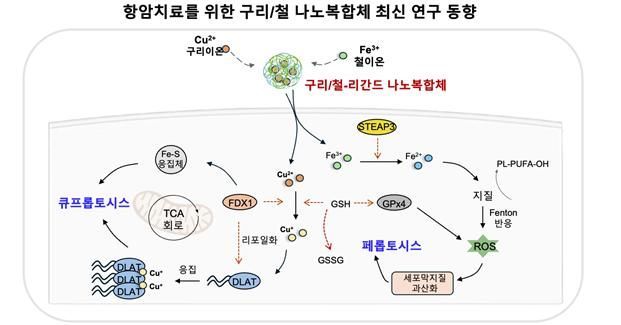
Schematic Diagram of the Principle of Anticancer Treatment. Kyung Hee University
View original imageTraditional anticancer drugs primarily induce apoptosis. However, many cancer cells develop resistance to this mechanism, which has limited the effectiveness of such treatments.
The research team led by Professor Do Kyung Kim focused on cuproptosis and ferroptosis. Cuproptosis is a form of cell death triggered by protein aggregation induced by copper ions, while ferroptosis is caused by enhanced lipid peroxidation promoted by iron ions.
These mechanisms are fundamentally different from existing treatment approaches. The research team summarized the structural characteristics and mechanisms of various copper- and iron-based nanocomposites.
Through this, they emphasized that simultaneously activating both pathways within cancer cells can produce a complementary and powerful anticancer effect.
When copper/iron-ligand nanocomposites are delivered to cancer cells, they release copper (Cu2+) and iron (Fe3+) ions. The released copper ions cause excessive aggregation of the DLAT protein in the mitochondrial TCA cycle, leading to copper-dependent cell death (cuproptosis).
At the same time, the iron ions induce lipid peroxidation by amplifying the generation of reactive oxygen species (ROS) via the Fenton reaction, thereby triggering iron-dependent cell death (ferroptosis).
The research team proposed that a single nanocomposite can simultaneously activate both cell death pathways, thereby maximizing therapeutic efficacy.
Professor Do Kyung Kim's latest paper goes beyond introducing basic research; it presents a strategy for precisely controlling the stability, redox reactivity, and tumor-specific metal ion release of nanocomposites through 'ligand engineering.' This marks an important milestone for the development of next-generation anticancer nanomedicines based on precision medicine.
Professor Do Kyung Kim stated, "Our findings demonstrate that copper- and iron-based nanoplatforms can serve as new weapons to overcome drug resistance in cancer cells," adding, "Further development of diverse materials, translational research from basic to clinical studies, and global collaboration will be needed for future clinical applications." Co-corresponding author, Research Professor Byung Seok Cha, added, "This paper introduces a new horizon in cancer treatment and will significantly contribute to the development of personalized anticancer therapies based on nanomedicine."