"Producing Gasoline Without Oil"... Demonstration of 50 kg Daily Production from Carbon Dioxide [Reading Science]
by Kim Jonghwa
Published 28 Apr.2026 12:00(KST)
Direct Conversion Without Intermediates
Initiating Design of a Commercial Process for Over 100,000 Tons Per Year
A domestic research team has advanced the technology to directly convert carbon dioxide (CO₂) into liquid fuels such as gasoline and naphtha, elevating it to the demonstration stage. Unlike conventional methods that use carbon monoxide (CO) as an intermediate, this approach enables a one-step conversion, simultaneously reducing both energy consumption and process complexity.
On April 28, a research team led by Dr. Kim Jeongrang at the Korea Research Institute of Chemical Technology, in collaboration with GS Engineering & Construction and Hanwha TotalEnergies Co., Ltd., announced that they have developed catalyst and process technology that directly reacts hydrogen (H₂) with carbon dioxide to produce liquid hydrocarbons, and succeeded in pilot production at a scale of 50 kg per day.
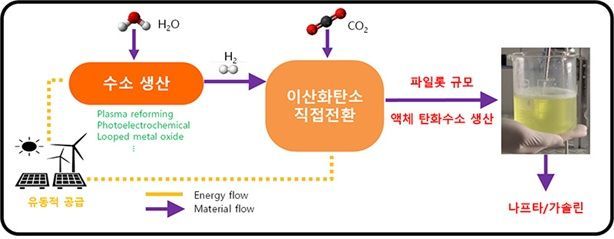
Schematic diagram of PtL technology that produces liquid hydrocarbons by converting carbon dioxide in connection with renewable energy. Provided by the research team
원본보기 아이콘This research was carried out with support from the Ministry of Science and ICT's "Carbon Resource Platform Compounds Research Group" project. Previously, the team had transferred 5 kg per day technology to the industry, and by the end of 2025, they plan to expand to the demonstration phase by building a 50 kg-scale pilot plant. They are also planning to initiate the design of a commercial process capable of producing more than 100,000 tons annually.
Overcoming the Limitations of High-Temperature, Multi-Step Processes... Implementation of a 300℃ Single-Step Reaction
The conventional process is a two-step system in which carbon dioxide is first converted to carbon monoxide, which is then synthesized into liquid fuels. This requires high-temperature reactions above 800℃ (reverse water-gas shift reaction, RWGS) and additional reactors, resulting in complex equipment and high energy consumption.
In contrast, the new technology uses a single-catalyst process in which carbon dioxide and hydrogen directly react at around 300℃ and 20 bar of pressure to generate liquid hydrocarbons. The research team explained that simplifying the process reduces equipment burden and improves energy efficiency. By combining multi-step reactions with recycling processes, they achieved a synthesis yield of approximately 50%.
The team established a foundation for long-term operation by improving catalyst manufacturing and operating conditions, and also demonstrated the potential for reducing production costs.
This technology is evaluated as a carbon resource technology capable of transforming the raw material structure of the refining and petrochemical industries. It enables the production of not only gasoline for automobiles but also naphtha, a key feedstock for plastics and synthetic fibers, from carbon dioxide.

Researchers at the Korea Research Institute of Chemical Technology. From the top right corner, moving counterclockwise: Senior Researcher Hyungki Min, Principal Researcher Haegoo Park, Principal Researcher Jeongrang Kim, Researcher Minjun Park, Researcher Taejung Lee, Khasan Nasriddinov, KRICT-UST Student Researcher, Researcher Aeri Kim, Postdoctoral Researcher Jingyu Chen, Student Researcher Suhyun Ryu, Researcher Jieun Min. Courtesy of KRICT
원본보기 아이콘In particular, it is significant as it allows carbon dioxide emitted from power plants and factories to be reused as raw material, targeting both carbon neutrality and energy security. This is drawing attention as an alternative raw material technology amid growing uncertainty in crude oil supply.
The research team stated, "If combined with hydrogen produced from renewable energy, this will become a core element of Power-to-Liquids (PtL) technology that converts electricity into liquid fuels."
However, there are still challenges to commercialization, including verifying catalyst lifespan and long-term stability, maintaining performance in real emission gas environments, and reducing hydrogen supply costs.
The team aims to accumulate data and conduct economic analysis through long-term operation of the pilot plant, with the goal of commercial demonstration around 2030 and full-scale commercialization in the early 2030s.
This research achievement was published as the cover article in the March 2026 issue of the international journal "ACS Sustainable Chemistry & Engineering." The corresponding author is Dr. Min Hyungki of Korea Research Institute of Chemical Technology, and the first author is Postdoctoral Researcher Chen Jingyu.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.