"Immune-Activating 'Viral DNA Barcode' Discovered: Unveiling the Secret of Herpes Infection Response [Reading Science]"
by Kim Jonghwa
Published 27 Apr.2026 09:29(KST)
UNIST Finds poly(T) Repeat Sequence Activates Immune Sensor AIM2
New Clue for Infection Suppression and Treatment Strategies
Published in the International Journal Nature Communications
The operating principle of the body's "immune switch," which detects viruses, has been elucidated at the DNA level. For the first time, it has been confirmed that specific repeat sequences hidden within the viral genome act as a "barcode" that triggers the immune response. This discovery is drawing attention as a clue that could change the direction of future infectious disease treatment strategies.
On April 27, the Ulsan National Institute of Science and Technology (UNIST) announced that the research team led by Professor Lee Sangjun in the Department of Biological Sciences had identified that the 'poly(T)' repeat sequence present in herpesvirus DNA activates AIM2, an innate immune sensor, thereby inducing an inflammatory response and cell death in infected cells. This study was conducted in collaboration with Sungkyunkwan University, Jeju National University, and the Korea Virus Research Institute at the Institute for Basic Science (IBS). The results were published in the international academic journal Nature Communications on April 13.
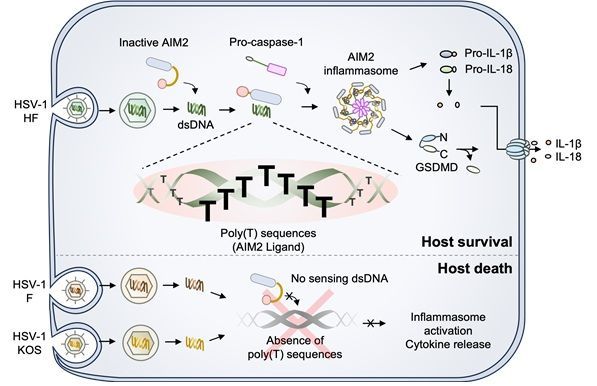
The principle of how the host's immune sensor recognizes viral DNA. The innate immune sensor AIM2 within host cells initiates an immune response by recognizing thymine repeat sequences specifically present in the herpes simplex virus type 1 (HSV-1) strain HF, thereby inducing the activation of inflammatory cytokines and cell death. Provided by the research team
원본보기 아이콘Herpes simplex virus type 1 is a common DNA virus, with approximately 67% of the global population infected. It typically remains dormant in nerve ganglia and reactivates when the immune system is weakened, causing inflammation around the lips. During this process, the host cell's innate immune system is activated, with the AIM2 protein playing a key role.
According to the research team, AIM2 recognizes regions of viral DNA where thymine (T) bases are repeated (poly(T)) to detect infection. In particular, even among the same herpesvirus species, the presence or absence of this repeat sequence led to vastly different immune responses. Strains containing the poly(T) sequence triggered strong inflammatory responses and cell death, whereas strains lacking this sequence or with a shorter sequence showed little to no immune response.
The longer the repeat sequence, the stronger the immune response: "Even affects survival rate"
The research team directly verified the function of this sequence through genetic manipulation. When the poly(T) region was removed, the immune response disappeared; conversely, inserting it into strains that originally lacked it induced a new immune response. The team also confirmed "length dependency," where the immune response became stronger as the repeat sequence grew longer.
The difference was also evident in animal experiments. When infected with viruses containing the poly(T) sequence, the immune response was activated and viral replication was suppressed. However, when the sequence was removed, there was little immune response, resulting in fatal infections. This demonstrates that even minor differences in the viral genome can determine the host's survival.

Photo of the research team. From the left: Professor Lee Sangjun, Researcher Oh Suhyeon, Researcher Oh Jueun, Researcher Lee Jihye. Provided by UNIST
원본보기 아이콘The research team also confirmed that this repeat sequence is commonly found not only in herpesviruses but also in a variety of viruses, including mpox virus, adenovirus, and coronavirus.
Professor Lee Sangjun explained, "We found that the human immune sensor recognizes viruses with much greater precision than previously thought. Rather than attacking viruses directly, this provides a theoretical foundation for developing new treatment strategies that modulate the activity of immune sensors." He added, "Since this repeat sequence is conserved among various infectious disease viruses, identifying the relationship between disease severity and genomic sequences will enable the design of more precise treatment strategies."
This research is significant in that it moves beyond conventional immune studies focused on viral proteins and demonstrates that the genome itself is a key determinant of the immune response. Experts are paying attention to the potential for this to lead to the development of "immune-modulating therapeutics" to respond to emerging and re-emerging infectious diseases in the future.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.