Patient Data Floods ASCO... Spotlight on Korean Biotech Clinical Results
by Park Jeongyeon
Published 27 Apr.2026 09:15(KST)
Vaizensell and GI Innovation to Deliver Oral Presentations
Immunoncia, Tiombio, and STCube to Present via Poster Sessions
Company Valuations Face a Test Amid Samchundang Fallout and Clinical Results
At least five domestic companies will simultaneously release clinical trial results from actual patients at the American Society of Clinical Oncology Annual Meeting (ASCO 2026), which will be held in Chicago, United States next month.
Earlier this year, at the JP Morgan Healthcare Conference-a major forum for technology transfer negotiations-Korean companies actively engaged in partnership discussions with global big pharma firms, but these did not result in large-scale deals. This is why the industry is focusing on the upcoming ASCO, which serves as an opportunity to prepare new cards for the next round of negotiations.
According to the pharmaceutical and biotech industry on April 27, this year's ASCO is particularly important due to fundamental changes underway in the global technology transfer market. Recently, global big pharma companies have shifted their strategies. Unlike in the past, when they would sign deals based solely on the potential of preclinical (animal testing) or early-stage clinical candidates, they are now focusing their investments on late-stage clinical pipelines where efficacy has been demonstrated in real patients.
According to a report published by Samil PwC Management Research Institute in December last year, the number of technology transfer deals for new drug discovery candidates in the first half of 2025 decreased by 10% compared to the same period the previous year. While the number of deals declined, the average deal size increased by about 80%. As big pharma’s standards have shifted to late-stage assets close to approval and market launch, the quality of the data presented by domestic companies-many of which are still in early clinical stages-at this ASCO will be the first hurdle in determining whether they can make it to the negotiation table for future technology transfer deals.
From Cell Therapy to Immuno-oncology... Patient Data Put to the Global Test
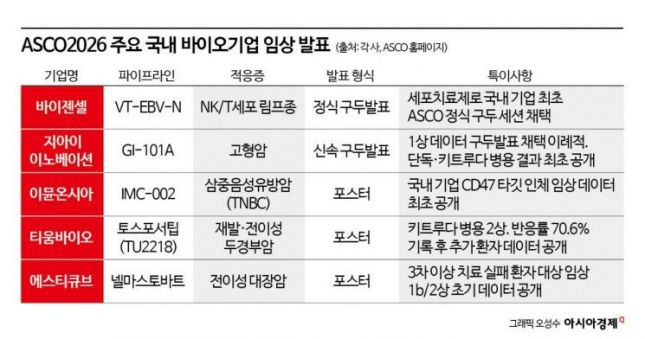
Vaizensell, which recently drew attention after receiving Korea’s first approval for an advanced regenerative medicine treatment plan, will present the results of its phase 2 trial (the second clinical trial in humans) for its cell therapy VT-EBV-N through an official oral presentation. This marks the first time a Korean company will present cell therapy clinical data in this session. VT-EBV-N is a therapy that uses a patient's immune cells to attack cancer cells in patients with the rare blood cancer NK/T-cell lymphoma, a cancer known for its high recurrence rate even after initial treatment. The key questions are how much VT-EBV-N suppresses recurrence and whether it extends overall survival.
GI Innovation’s immuno-oncology therapeutic GI-101A, which has been in combination trials with big pharma, was also selected for a rapid oral presentation. The fact that it was chosen based solely on phase 1 data is considered unusual. Results from both monotherapy and combination therapy with the immuno-oncology drug Keytruda (ingredient: pembrolizumab) in solid tumor patients will be simultaneously presented. Since GI Innovation licensed out the China rights to GI-101 in 2021 in a deal worth about 900 billion won but has not secured any subsequent deals, attention is focused on whether this new data can pave the way for new negotiations.
Immunoncia, a subsidiary of Yuhan Corporation, will present interim phase 1b results for its immuno-oncology antibody IMC-002 in triple-negative breast cancer (TNBC) via poster. IMC-002 is an antibody therapy that blocks the 'CD47' signal, which cancer cells emit to evade immune cell attacks, so that immune cells can once again attack the cancer. Since major big pharmas like Gilead and Pfizer have halted development of this target due to blood-related side effects, the real test will be whether Immunoncia can show differentiated safety results. This is also the first time a Korean company will present TNBC clinical data for a CD47-targeted therapy.
Tiombio, which attracted attention last year with interim data presented at the Society for Immunotherapy of Cancer (SITC) annual meeting in the United States, will present updated results from a phase 2a trial of its oral immuno-oncology drug Tosfosetib (TU2218) in combination with Keytruda via poster. The indication is recurrent or metastatic head and neck cancer. In November 2023, interim data showed an objective response rate (ORR) of 70.6%-about double the current standard therapy (36%)-garnering significant attention. This time, long-term follow-up data including additional patients will be released. Since anti-cancer drugs often see tumor shrinkage initially but regrowth over time, the key will be how long the response lasts, which will determine the real value of the drug.
STCube, which completed patient enrollment for its phase 2 trial in January this year, will present initial data from its phase 1b/2 trial of the immuno-oncology drug nelmastobart in metastatic colorectal cancer via poster. Colorectal cancer is the third most common cancer in Korea, but only about 5% of patients respond well to immuno-oncology drugs. The remaining 95%, classified as microsatellite-stable (MSS) types, are considered a blind spot as they rarely respond to existing immuno-oncology drugs. Nelmastobart targets a different molecule (BTN1A1) to address this area. The key focus of this announcement will be whether tumor suppression was achieved in patients who failed three or more prior therapies.
An executive from one of the biotech companies presenting clinical results at this year’s ASCO stated, “In the past, even having a plausible mechanism of action could spark technology transfer discussions, but now, without patient data, it’s nearly impossible to get a meeting with big pharma. This year’s conference will illustrate how the quality of data determines the quality of potential deals.”
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.