Celltrion's High-Margin New Products Drive Rapid Growth... Securities Firms See 31% Upside Potential
by Jeong Donghoon
Published 24 Apr.2026 06:30(KST)
Updated 24 Apr.2026 08:58(KST)
Celltrion Fills the Biosimilar Gap
Expanding Portfolio: 18 Products by 2030, 41 by 2038
Structural Benefits from U.S. Tariff Exemptions and Healthcare Policies
High Growth Expected from CMO Sales and More
Celltrion is expanding its influence in the global market by successfully launching new biosimilars and broadening its product portfolio. According to the securities industry, new products are expected to quickly gain market share starting this year, leading Celltrion into a phase of substantial performance growth.
"Rising Proportion of New Product Sales... Additional Revenue Expected"
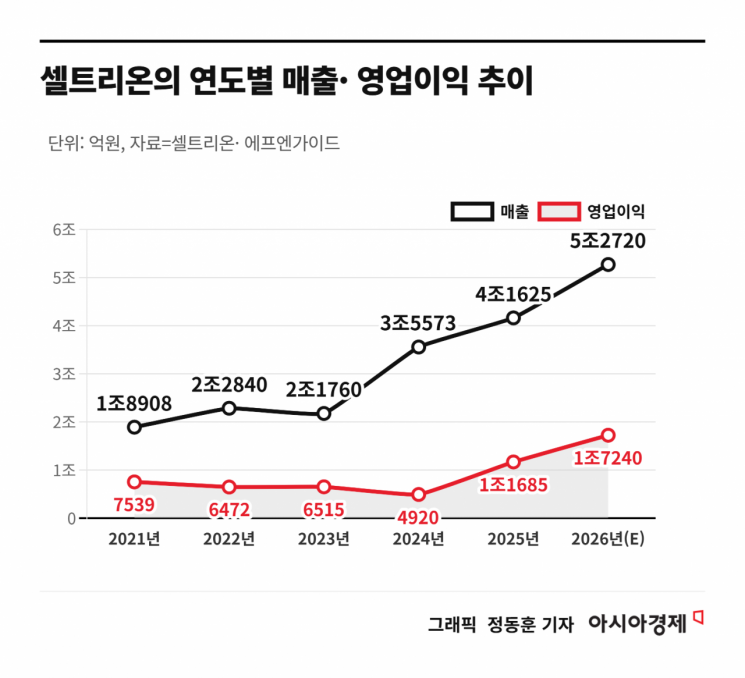
On April 24, sources in the pharmaceutical, biotech, and securities industries reported that 26 analysts from domestic and international securities firms unanimously gave Celltrion a "buy" rating. The average target price set by these analysts was around 2.7 million won, indicating an upside potential of more than 31% compared to the closing price of 2,060,000 won the previous day (April 23). Last year, Celltrion achieved a historic milestone, recording annual sales of 4 trillion won and operating profit of 1 trillion won for the first time. Notably, sales from highly profitable new products accounted for 54% of total revenue, surpassing half and significantly driving performance growth.
Kim Minjeong, a researcher at DS Investment & Securities, stated, "Thanks to the growth of follow-up pipelines such as Omriclo and Stoboclo, we expect Celltrion to continue its explosive growth this year, reaching annual sales of approximately 5.3476 trillion won and operating profit of about 1.7269 trillion won." Heo Hyemin, an analyst at Kiwoom Securities, added, "The proportion of sales from high-margin new products will expand from 54% last year to 66% this year,” and analyzed that “new products, including Omriclo and the planned U.S. launch of Idengelt in the second half of this year, will drive performance growth.”
Jung Jaewon, a researcher at IM Investment & Securities, also commented, "The improvement in performance due to the increasing share of new products in the core biosimilar business has continued since the second half of last year," and added, "This year, we expect to secure additional revenue through expanded sales regions for new products."
Number of Biosimilars to Expand from 11 to 18 to 41... Poised to Become a Strong Cash Cow
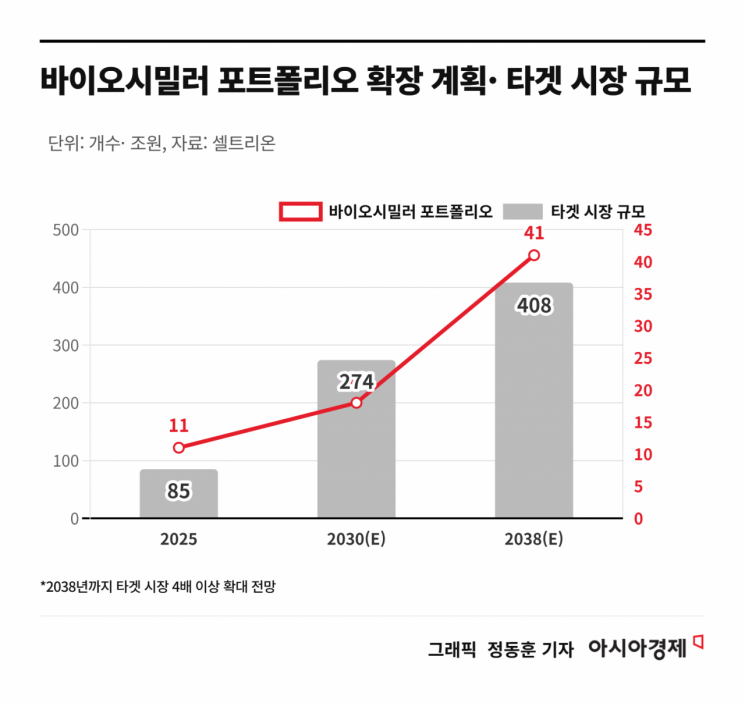
The core drivers behind this growth are the expansion of the product lineup and sustained investment in research and development. Last year, Celltrion invested a record-breaking 482.4 billion won in R&D, accelerating new product launches. The company launched 5 new products last year alone, bringing its total to 11 products and significantly expanding its business scale. Celltrion plans to expand its portfolio from the current 11 products to 18 by 2030 and 41 by 2038.
The company is also diversifying its target disease areas. While maintaining a strong presence in its traditional strengths-autoimmune diseases and oncology-it is gradually broadening its pipeline to include allergies, ophthalmology, and bone diseases. Based on its 11 current products, the addressable market size is around 85 trillion won, which is expected to expand to about 274 trillion won by 2030 and 408 trillion won by 2038, forming a vast mega market. The synergy between legacy and new products is projected to make the biosimilar business a long-term cash generator.
Beyond its core biosimilar business, the expansion of its pharmaceutical contract manufacturing operations is also serving as a new growth engine and contributing to increased profitability. Earlier this year, Celltrion signed a large-scale contract manufacturing agreement worth about 678.7 billion won with multinational pharmaceutical company Eli Lilly, followed by another deal with a different global pharmaceutical firm amounting to up to 375.4 billion won last month. As a result, the company exceeded 1 trillion won in cumulative order intake in just the first quarter of this year. These orders will be gradually reflected in sales, contributing to improved profitability. The company plans to further increase contract manufacturing orders leveraging its own production capabilities, positioning this as a key new growth driver.
The business environment in the United States-the world’s largest pharmaceutical market-has also become increasingly favorable for Celltrion. In the short term, the company has secured a stable supply of its products in the U.S., and in the mid- to long-term, it has established a system to produce and supply products directly from its manufacturing facility in Branchburg, New Jersey, thereby avoiding tariff risks.
The U.S. government’s healthcare cost reduction policies also bring structural benefits. The government has excluded biosimilars, which account for most of Celltrion’s U.S. sales, from tariffs. In addition, policies have been announced to increase the financial burden on private insurers operating Medicare for the elderly and raise the cap on patients’ out-of-pocket expenses. These measures are designed to naturally promote prescriptions of lower-priced biosimilars and ensure the sustainability of healthcare finances, which is expected to accelerate Celltrion’s market penetration.
A Celltrion official commented, "As new biosimilar products are successfully settling in major global markets, our overall revenue structure is taking a significant leap forward. This year, we will continue to pursue solid performance growth centered on highly profitable new products, focus on expanding our product portfolio over the mid- to long-term, and capitalize on favorable policy environments in key countries to position ourselves as a leading global pharmaceutical company."
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.