CGBio's Patient-Specific Titanium Implant 'EasyMade TI' Receives FDA 510(k) Clearance in the U.S.
by Jeong Donghoon
Published 22 Apr.2026 08:58(KST)
CGBio announced on April 22 that its patient-specific titanium implant, 'EasyMade TI (EASYMADE TI),' received 510(k) clearance from the U.S. Food and Drug Administration (FDA) on April 9. The FDA 510(k) is a representative premarket approval process in the United States, which requires proof of substantial equivalence to an already authorized product for medical device sales within the country.
This achievement marks the first time a Korean company has obtained FDA 510(k) clearance for a patient-specific titanium implant intended for the U.S. market. It is highly significant as it demonstrates both CGBio's technological capability and its proficiency in navigating global regulatory requirements. In particular, it is regarded as an example of a Korean company's competitiveness in the field of patient-specific cranial and craniofacial reconstruction solutions.
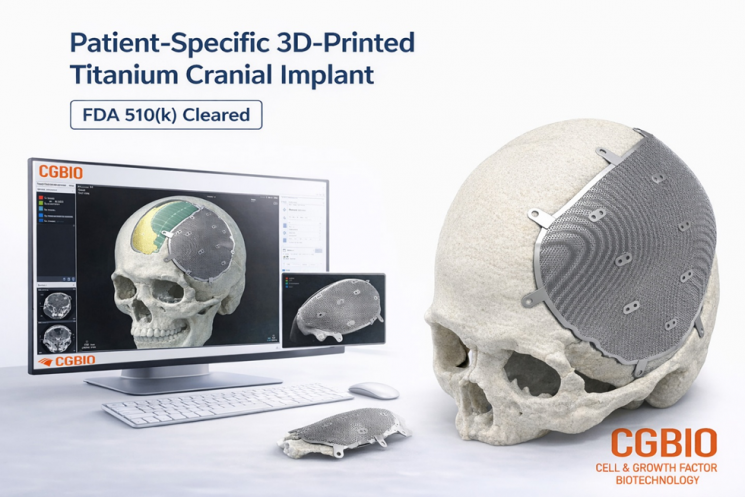
EasyMade TI is a patient-specific titanium implant designed based on a patient's CT data, developed for the reconstruction of cranial and non-load-bearing craniofacial defects. The product uses medical-grade titanium alloy and is manufactured using the Laser Powder Bed Fusion (LPBF) process, a metal 3D printing technology.
The product supply process is also noted as a competitive advantage. When U.S.-based medical professionals send a patient's CT images, CGBio's design center in Korea creates a customized design and then ships the product to the U.S. hospital within five days. After undergoing sterilization at the hospital, the implant can be immediately used in surgery.
This clearance is expected to serve as a turning point for CGBio's expansion of its customized implant business in the United States. In the U.S. market, demand for reconstruction is steadily increasing following trauma, tumor resection surgery, and decompressive craniectomy. There is also growing interest in customized reconstruction solutions optimized for each patient's anatomical structure. CGBio plans to accelerate its efforts to capture the cranial and craniofacial reconstruction market in the United States in response to this expanding demand.
Hyunseung Yoo, CEO of CGBio, said, "The FDA 510(k) clearance for EasyMade TI is a meaningful achievement that demonstrates CGBio's global competitiveness in the field of patient-specific reconstruction solutions," adding, "With this approval, we will actively expand our customized cranial and craniofacial reconstruction implant business in the U.S. market and proactively pursue global regulatory approval and commercialization of subsequent product lines."
Building on this clearance, CGBio will begin full-scale commercialization in the United States and continue to expand its global presence in the field of patient-specific medical devices.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.