The Second Act of K-Beauty Is a 'Tech War'... Aiming to Lead the Beauty Medical Device Market
by Kwon Jaehee
Published 11 Mar.2026 07:14(KST)
Home Beauty Device Market Grows at Over 10% Annually
Recurring Purchases Enable Subscription-Based Revenue
The domestic cosmetics industry, which has fueled the global K-beauty phenomenon, is now poised to compete in the beauty medical device market. After evolving beyond cosmetics manufacturing and sales to introduce home beauty devices, companies are now expanding into the beauty medical device sector, effectively preparing for the 'second act' of K-beauty.
While the cosmetics market has entered a mature stage and faces intensified competition, making repeat purchases difficult, the home beauty device and medical device markets are expected to experience high annual growth rates of over 10%. This is driven by the recurring purchase of consumables, such as cartridges and ampoules, rather than the devices themselves.
According to the cosmetics industry on March 11, APR recently announced that it would amend its articles of incorporation at a shareholders' meeting to include medical device-related business purposes. The new business purposes include "development, manufacturing, and sales of medical equipment" and "medical device repair business." The market interprets this not merely as a business scope expansion but as a preparatory move to enter the medical device industry.
Currently, most home beauty devices available in the market are intended for skincare or beauty purposes. However, to offer features for medical purposes-such as acne treatment, scar improvement, skin regeneration, or hair loss management-a company must obtain a medical device manufacturing license and specify related business purposes in its articles of incorporation. This is why APR's move to amend its articles is seen as laying the institutional groundwork for future medical device certification.
Amorepacific also showcased its commitment to technology-based beauty services at this year’s Consumer Electronics Show (CES) by unveiling an "AI skin analysis and care solution" in collaboration with Samsung Electronics. At the same time, through its brand "MakeON," the company introduced a variety of beauty device products, signaling its intent to target the digital beauty market.
Although Amorepacific launched "MakeON" back in 2014, it failed to achieve significant results due to the lack of widespread adoption of beauty devices. Starting this year, however, the company is focusing on home beauty devices as a core business and plans to introduce beauty devices and dedicated cosmetics through "MakeON" in the second half of the year. Next year, it aims to expand this business to hair and scalp care as well.
Cosmetics ODM (Original Design Manufacturing) companies are also responding quickly. Kolmar Korea unveiled its "SKA Beauty Device" at this year's CES. Cosmax also demonstrated its presence in the technology-driven beauty market, with "Maxface" winning an innovation award in the beauty tech category at CES this year. This device analyzes users' skin condition, humidity, and lifestyle data in real time to recommend and combine optimal cosmetic formulations. The data generated as consumers use these products is then utilized to design new cosmetics.
The reason traditional cosmetics companies are turning their attention to beauty devices and medical device sectors is closely tied to changes in industry structure. While the traditional cosmetics (manufacturing) market is already mature, the home beauty device and medical beauty device markets continue to show high growth potential.
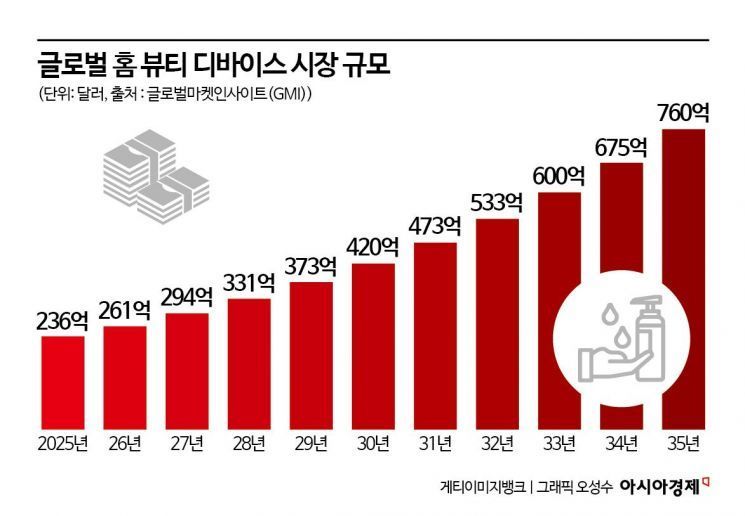
According to global market research firm Global Market Insights (GMI), the home beauty device market was estimated at approximately $23.6 billion (about 3.5 trillion won) last year and is expected to reach $26.1 billion (about 3.9 trillion won) this year. By 2035, the market is projected to grow to $76 billion (about 11.3 trillion won).
This market is particularly attractive because, unlike cosmetics, it enables recurring revenue streams. After purchasing a device, consumers must continuously buy consumables such as cartridges, ampoules, and dedicated gels. This creates a "lock-in effect," encouraging continued use of specific brands’ products and potentially leading to subscription services and regular purchases-a point of great interest for the industry.
The pharmaceutical and biotech sectors are also expanding into the beauty device market. Pharmaresearch, a regenerative medicine-based company, is leveraging its "Rejuran" technology-known for skin regeneration procedures-to expand into the home care product and device sectors. DongKook Pharmaceutical has also entered the home beauty market by introducing the "Madeca Prime" skincare device under its dermacosmetic brand "Centellian 24." Classys, a medical beauty device company, has launched the home-use "Shurink Home" device, applying the ultrasound lifting technology of its hospital-grade "Shurink" equipment, to target consumers directly.
A cosmetics industry official commented, "Once consumers purchase a device, the structure compels them to continue using consumables, which is a key point for companies to secure stable, recurring revenue. If companies design subscription-based models around this, they can strategically lock consumers into their brand ecosystem."
Kim Jiyeong, a researcher at Shinhan Investment Corp., analyzed, "The global beauty medical industry has entered a phase of rapid expansion, with the total addressable market size growing quickly. As consumer selection criteria are rapidly shifting toward products that simultaneously offer price rationality, safety, and technological sophistication, domestic beauty medical device companies are well-positioned to capture global demand."
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.