KAIST and Seoul National University Present New Standard for Greenhouse Gas Reduction Catalyst Design
by Jeong Ilwoong
Published 04 Feb.2026 08:35(KST)
A study has found that the effectiveness of greenhouse gas reduction varies depending on how catalytic technology is used. By revealing the principle that oxygen can be selectively utilized according to the environment in which the catalyst operates, the researchers are being credited with presenting a new standard for catalyst design.
KAIST announced on the 4th that a joint research team, including Professor Lee Hyunju of the Department of Chemical and Biomolecular Engineering at KAIST, Professor Park Jungyoung of the Department of Chemistry at KAIST, and Professor Han Jungwoo of Seoul National University, has for the first time in the world identified that the way oxygen is used changes completely depending on the size of ceria (CeO₂).

(from bottom left) Professor Lee Hyunju, PhD candidate Choi Yoonji, PhD candidate Han Jaebeom, Professor Park Jeongyoung, (from top left) Professor Han Jungwoo, Dr. Jung Seokhyun. KAIST
원본보기 아이콘Ceria is an eco-friendly metal oxide catalyst that replaces or supplements expensive precious-metal catalysts. Because it can store oxygen and release it when needed, it is also called an "oxygen tank" in the catalyst field. However, it has not been clearly understood where the oxygen stored in ceria is accumulated or under what conditions it reacts.
The joint research team approached the characteristics of ceria not simply as a "catalyst that uses oxygen well," but as a "catalyst that selectively uses oxygen depending on the situation." They manufactured catalysts in which ceria was precisely controlled from the nanoscale to relatively large sizes, and then systematically analyzed the processes of oxygen migration and reaction.
As a result, they confirmed that small-sized ceria catalysts operate in a "quick-response mode," rapidly taking up oxygen from the air and using it immediately in reactions, whereas large-sized ceria catalysts play an "endurance mode" role, continuously supplying oxygen by drawing the oxygen stored inside up to the surface.
In other words, they have revealed a design principle by which one can choose, solely by adjusting the size of the catalyst, "whether to use oxygen from the air or oxygen stored inside the catalyst, depending on the reaction conditions." This mechanism was simultaneously verified through advanced experimental analysis and artificial intelligence-based simulations.
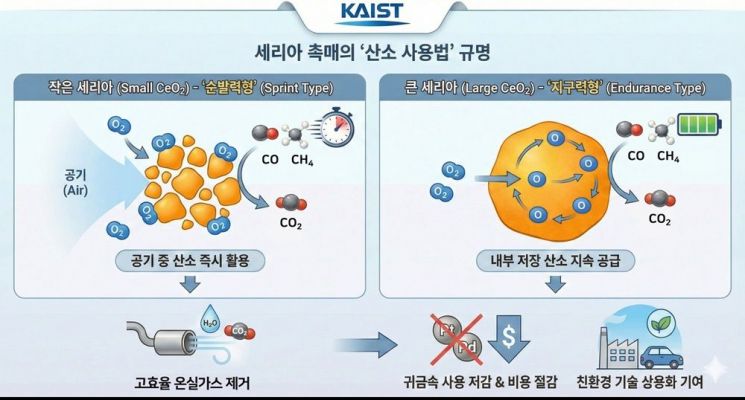
Image of ceria catalyst application (AI-generated). Korea Advanced Institute of Science and Technology
원본보기 아이콘The joint research team also applied this principle to methane removal experiments. Methane is a greenhouse gas with a warming effect tens of times stronger than that of carbon dioxide. It can be removed through a catalytic oxidation reaction that uses oxygen to convert methane into carbon dioxide and water.
In the experiments, the small ceria catalyst demonstrated stable methane removal performance even at low temperatures and in highly humid environments by immediately utilizing oxygen from the air. This shows that the amount of expensive precious-metal catalysts such as platinum and palladium can be reduced while still improving performance.
This achievement is expected to help reduce the manufacturing cost of environmental purification equipment and to lead to the development of highly durable catalysts whose performance is maintained even under real industrial conditions such as rain and high humidity, thereby accelerating the commercialization of eco-friendly energy and environmental industries.
Professor Lee Hyunju said, "This study is meaningful in that it clearly distinguishes, for the first time, the two key mechanisms by which oxygen operates in catalysts," adding, "It has opened a new path to 'custom-design' high-efficiency catalysts tailored to the reaction conditions needed to respond to the climate crisis."
Meanwhile, this research included KAIST PhD candidate Choi Yoonji from the Department of Chemical and Biomolecular Engineering, Dr. Jung Seokhyun from the Department of Materials Science and Engineering at Seoul National University, and KAIST PhD candidate Han Jaebeom from the Department of Chemistry as co-first authors. The research results (paper) were published in the international journal Nature Communications on the 9th of last month.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.