Obesity Drug Battle: Mounjaro Scores a Decisive Victory in the U.S.
by Park Jeongyeon
Published 26 Jan.2026 08:47(KST)
Mounjaro Rises to Top Prescription Spot in the US and Korea
Oral Obesity Medications Gain Attention Amid Competition for Dosing Convenience
Competition Intensifies for Next-Generation Obesity Treatments
In the United States, the largest market for obesity treatments, Eli Lilly's Mounjaro (ingredient: tirzepatide), a latecomer to the field, has surpassed Novo Nordisk's Wegovy (ingredient: semaglutide). Recently, Mounjaro's number of prescriptions in South Korea also exceeded that of Wegovy, and now, in the global market, Mounjaro is establishing itself as the industry’s leading product.
Wegovy, which pioneered the glucagon-like peptide-1 (GLP-1) obesity treatment market and led the way with high public recognition, initially secured an advantage. However, as the perception spread that its weight loss effects lag behind those of Mounjaro, it has lost its dominance.
As the obesity treatment market enters a phase of long-term therapy, global pharmaceutical companies are shifting their competition away from simple market preemption toward a direct contest over differences in weight loss efficacy and improvements in dosing and administration convenience.
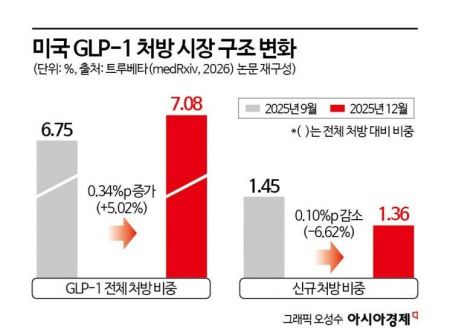
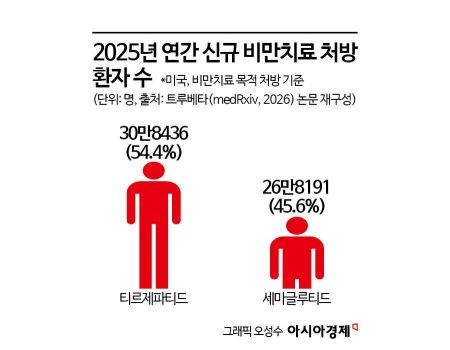
According to research results released on January 22 (local time) by the Truveta research team, a U.S. medical data analytics company, on the preprint site 'medRxiv,' the number of new patients prescribed obesity treatments in the United States in 2025 is projected to be 308,436 for tirzepatide, surpassing 268,191 for semaglutide. According to the research team, this is the first time the rankings of the two drugs have been reversed based on annual new prescriptions. The year 2025 is thus seen as the first year in which Mounjaro’s dominance in the market is firmly established.
Mounjaro's superiority is also clear in the Korean market. Within just four months of its domestic launch, Mounjaro approached 100,000 prescriptions, overtaking the previously leading Wegovy. According to data submitted by the Health Insurance Review and Assessment Service to Assemblywoman Seo Mihwa of the National Assembly’s Health and Welfare Committee, Mounjaro prescriptions in November last year reached approximately 97,000 cases, an increase of more than 23% compared to the previous month. This figure is more than five times higher than in August last year, the first month of its launch. Meanwhile, Wegovy prescriptions peaked in September last year and declined for two consecutive months.
Competition in the GLP-1 obesity treatment market is expected to enter a second phase, focusing on long-term administration convenience. According to this study, in the previous year, only 47.5% of those prescribed GLP-1 obesity treatments actually collected their medication within 60 days. This is interpreted as a sign that the obesity treatment market is shifting from the initial expansion phase to a structure centered on long-term users. In particular, for obesity indications, limited insurance coverage and high drug costs mean that prescriptions often do not translate into actual use. As a result, strategies to enhance patient convenience in administration while reducing cost burdens are emerging as key competitive factors for global pharmaceutical companies.
Competition over absolute superiority in weight loss efficacy is also expected to unfold simultaneously. In clinical trials conducted by Eli Lilly, high-dose administration of Mounjaro resulted in an average weight loss rate of about 21%, surpassing the main clinical results for Wegovy (about 15%). As these results became known, patient preference for Mounjaro rapidly increased. Currently, research is accelerating on triple-action mechanisms, which are a step beyond the dual-action GLP-1 and glucose-dependent insulinotropic polypeptide (GIP) agonist approach adopted by Mounjaro. As such, the battle for global market leadership is expected to become even more intense.
To reclaim its top position, Novo Nordisk is countering with oral medications. In an effort to disrupt the market structure dominated by once-weekly injections, the company is working to expand the obesity indication for oral semaglutide, known as the "pill form of Wegovy." Oral semaglutide for obesity treatment received approval from the U.S. Food and Drug Administration (FDA) at the end of last year and will begin full-scale market entry early this year.
Novo Nordisk is also pursuing a combination mechanism strategy to enhance weight loss efficacy. The company is developing CagriSema, a combination candidate of semaglutide and an amylin agonist, as a next-generation obesity treatment. CagriSema combines the long-acting amylin analogue cagrilintide with the GLP-1 agonist semaglutide in a once-weekly injectable formulation. If approved, it will become the first GLP-1 and amylin combination obesity treatment.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.