Pharmicell Succeeds in Synthesizing PEG-Based Artificial Blood
Published 26 Aug.2025 10:15(KST)
Pharmicell announced on August 26 that it has succeeded in synthesizing Hempharmin-16, the world’s first fully synthetic artificial blood. This marks the first case in the 90-year global history of artificial blood development to simultaneously achieve safety, efficacy, and cost-effectiveness, and is being hailed as an innovation that could transform the industry landscape.
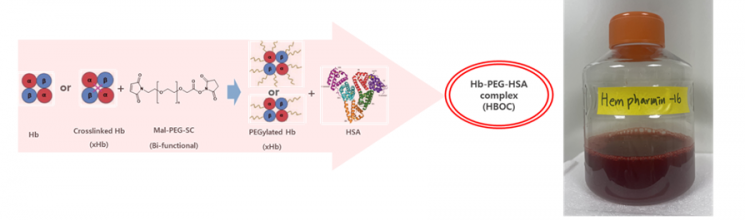
Hempharmin-16 is a next-generation artificial blood that, unlike conventional cell-based methods, directly synthesizes human body components and pharmaceutical ingredients to replicate the properties of real blood. In particular, Pharmicell applied its proprietary Mal-PEG-SC(5k) dual-function linker system, successfully stabilizing the molecular binding of hemoglobin and albumin. This fundamentally resolves side effects such as vasoconstriction and renal toxicity, which have been issues with existing products.
Through joint verification with Wonju Severance Christian Hospital, Pharmicell demonstrated Hempharmin-16’s performance: oxygen-carrying capacity of 97.1% (equivalent to normal blood at 96.6%), blood gas reactivity (CO₂/O₂ response identical to natural blood), and temperature stability (complete maintenance of electrolyte concentration from 4℃ to 40℃). This represents a dramatic improvement over existing artificial blood, which typically achieved only 60-80% performance levels.
The product meets both the functionality (over 90% of donor-derived stored blood) and safety standards for whole blood substitutes set by the U.S. Defense Advanced Research Projects Agency (DARPA). Pharmicell has completed international patent applications through domestic patents and the Patent Cooperation Treaty (PCT).
Hempharmin-16 is an innovative alternative that offers both versatility and cost-effectiveness, featuring over 97% oxygen-carrying capacity and biocompatibility compared to natural blood, reduced production costs and a stable supply chain due to the absence of cell culture, universal application regardless of blood type, and the potential for long-term preservation.
Kim Hyunsoo, CEO of Pharmicell, stated, “Hempharmin-16 is not just an incremental improvement, but a paradigm-shifting technology that ensures both safety and efficacy. We aim to lead the global market as a core solution that saves lives in various fields, including emergency medicine, treatment of massive hemorrhage, and remote medical care.”
Pharmicell is currently preparing for preclinical animal trials by enhancing large-scale synthesis and purification technologies as a step toward clinical trials. According to global market research firm Data Bridge Market Research, the global artificial blood market is projected to grow at an average annual rate of 20%, from $5.6 billion (approximately 7.6 trillion KRW) in 2021 to $24.08 billion (approximately 32.8 trillion KRW) in 2029.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.