Pukyong National University Research Team Discovers Anion Separation Phenomenon in All-Solid-State Batteries
Published 17 Dec.2024 08:10(KST)
Research by Professor Jeong Seong-cheol's Team Published in the Royal Society of Chemistry International Journal
Localized Anion Separation Phenomenon Causes High Ionic Conductivity
On the 16th, Pukyong National University (President Sang-Hoon Bae) announced that Professor Seongcheol Jeong's research team (Department of Physics) discovered a phenomenon where monovalent and divalent anions are locally separated in the Li6PS5Cl series solid electrolytes with an argyrodite structure used in all-solid-state batteries.
The research team led by Professor Seongcheol Jeong, postdoctoral researcher Seho Lee (G-LAMP Project Group), and Ph.D. candidate Taegon Jeon (Department of Physics) revealed through first-principles calculations that the difference in lithium mobility according to anion domains caused by local anion separation found in Li6-xPS5-xClBrx solid electrolytes is the reason for the high ionic conductivity of the Li6PS5Cl series solid electrolytes.

Research team (from left: Jeong Seong-cheol, Lee Se-ho, Jeon Tae-gon). Provided by National Pukyong University
원본보기 아이콘Solid electrolytes based on the argyrodite structure have excellent ionic conductivity, economic raw materials, and easy synthesis, leading to active related research.
In this study, Professor Jeong's team demonstrated that lithium ions are more distributed in the S2- divalent anion domains than in the Cl- and Br- monovalent anion domains, effectively stabilizing the argyrodite structure.
They also confirmed that lithium ions distributed in the divalent anion domains exhibit strong bonding with anions and thus show low mobility, whereas lithium ions in the monovalent anion domains have relatively weaker bonding with anions and exhibit high mobility. Through this, they elucidated that lithium mobility dependent on anion domains is the lithium diffusion mechanism in argyrodite solid electrolytes.
Professor Seongcheol Jeong said, "This study shows that the well-known disordered arrangement of anions in the Li6PS5Cl series argyrodite solid electrolytes is actually related to the local separation phenomenon of anions, providing a new perspective to understand the cause of the high conductivity of argyrodite solid electrolytes."
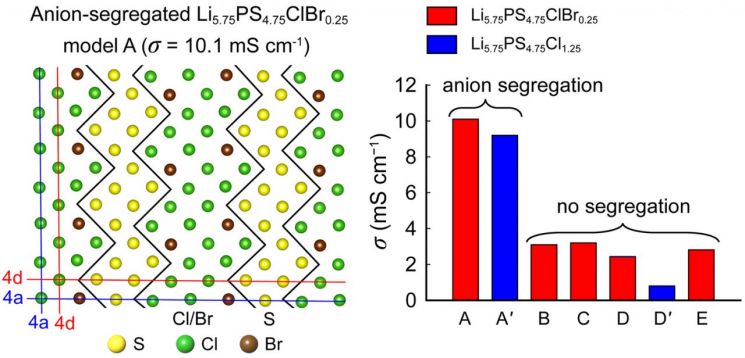
Graph of anion separation in Li6PS5Cl-based solid electrolytes and the resulting conductivity enhancement.
원본보기 아이콘This research was conducted with support from the Ministry of Education's G-LAMP project and was recently published in a prestigious international journal in the fields of chemistry, physics, and materials published by the Royal Society of Chemistry (IF=10.8).
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.