EGIYON "Solid Oxide Fuel Cell Performance Can Be Improved Threefold in '4 Minutes'"
by Jeong Ilwoong
Published 12 Jun.2024 15:24(KST)
A catalyst coating technology that can boost the performance of Solid Oxide Fuel Cells (SOFC) up to three times in just 4 minutes has been developed domestically.
The Korea Institute of Energy Research (KIER) announced on the 12th that Dr. Choi Yoon-seok from the Hydrogen Composite Materials Research Laboratory, in collaboration with Professor Jung Woo-cheol of the Department of Materials Science and Engineering at KAIST and Professor Park Beom-gyeong of the Department of Materials Engineering at Pusan National University, achieved this result through joint research.
Fuel cells are attracting attention as a high-efficiency clean energy source leading the hydrogen economy. Among them, SOFCs are fuel cells operating at high temperatures above 700 degrees Celsius, composed entirely of solid electrodes and electrolytes. They can use various fuels such as hydrogen, biogas, and natural gas, and enable combined power generation by utilizing heat generated during the process, making them a subject of active research.
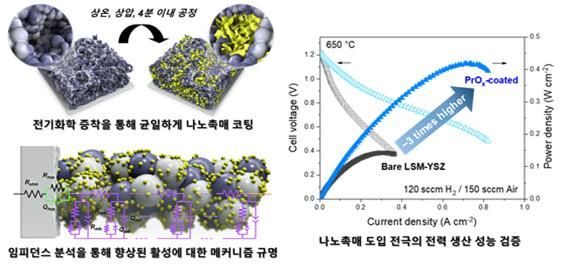
Schematic of a commercial composite electrode coated with nanocatalysts. Provided by Korea Institute of Energy Research
원본보기 아이콘The performance of SOFCs is determined by the oxygen reduction reaction occurring at the air electrode (cathode). Compared to the reactions at the fuel electrode (anode), the reaction rate at the air electrode is slower, limiting the overall reaction speed. Researchers have focused on developing new air electrode materials with high activity to overcome the slow speed of SOFCs, but further research was needed due to a lack of chemical stability.
The joint research team concentrated on enhancing the performance of the LSM-YSZ composite electrode (hereafter composite electrode), which is widely used in industry due to its excellent safety, to overcome these limitations.
As a result, they succeeded in developing a nano-sized praseodymium oxide (PrOx) catalyst coating process that actively promotes the oxygen reduction reaction on the surface of the composite electrode, enabling a significant improvement in SOFC performance when applied.
During the research process, the team introduced an electrochemical deposition method (a technique that deposits metals or metal compounds on the electrode surface using electrochemical reactions) that operates at room temperature and pressure without requiring complex equipment or processes.
By immersing the composite electrode in a solution containing praseodymium (Pr) ions and applying an electric current, hydroxide ions (OH-) generated on the electrode surface react with praseodymium ions to form a precipitate that uniformly coats the electrode.
The coating layer formed in this way undergoes a drying process to convert into an oxide form, which stably promotes the oxygen reduction reaction of the electrode even in high-temperature environments. The coating process takes only 4 minutes.

Researchers participating in the joint study. (From left) Nam Seong-woo, PhD student in the Department of Materials Science and Engineering at KAIST; Jin-wook Kim, PhD; Woo-cheol Jeong, Professor; Beom-kyung Park, Professor in the Department of Materials Science and Engineering at Pusan National University; Yoon-seok Choi, Senior Researcher at the Korea Institute of Energy Research. Provided by the Korea Institute of Energy Research.
원본보기 아이콘In particular, the joint research team elucidated the principle by which the coated nano catalyst facilitates oxygen exchange and ion conduction, providing academic evidence that the catalyst coating method can solve the low reaction rate of the composite electrode.
When comparing the composite electrode with the developed catalyst applied and the conventional composite electrode after operating for more than 400 hours, it was confirmed that the resistance generated during the electrochemical reaction was reduced by a factor of 10.
They also explained that the fuel cell with this application showed three times higher power generation performance (142mW/cm² → 418mW/cm²) than conventional fuel cells even at a low temperature of 650 degrees Celsius. This is among the highest levels of SOFC performance reported in academia using LSM-YSZ composite electrodes.
Co-corresponding author Dr. Choi Yoon-seok stated, “The electrochemical deposition technology developed this time is a post-processing step that does not significantly affect the existing SOFC manufacturing process,” and evaluated that “introducing oxide nano catalysts is economically advantageous and has high potential for widespread industrial use.”
He added, “The joint research team regards this study as significant because it secured core technology applicable not only to SOFCs but also to various energy conversion devices such as high-temperature water electrolysis (SOEC) for hydrogen production.”
Meanwhile, the joint research team conducted the study with support from the Ministry of Trade, Industry and Energy’s Renewable Energy Core Technology Development Project and the Ministry of Science and ICT’s Individual Basic Research Project. The research results were published in the world-renowned materials science journal ‘Advanced Materials.’
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.