'Rice of Industry' in the Eco-Friendly Era... Ethylene, Raw Material for Plastic and Vinyl, Produced from CO2 Instead of Oil
by Kim Yongu
Published 24 Apr.2022 11:59(KST)
Updated 10 Aug.2025 11:12(KST)
UNIST·KENTECH·KAIST Joint Research Team Develops Electrochemical Catalyst for CO2 to Ethylene Conversion
Records Highest Conversion Efficiency and Current Density Exceeding Commercialization Standards, Published in Leading Energy and Environment Jo

Photo of the developed copper-aluminum alloy catalyst observed with an electron microscope.
원본보기 아이콘[Asia Economy Yeongnam Reporting Headquarters Reporter Kim Yong-woo] Making ‘rice’ from carbon dioxide?
A technology that easily produces ethylene, known as the ‘rice of industry,’ in large quantities using carbon dioxide instead of petroleum has been developed in Korea, stirring excitement in the academic community.
Ethylene is considered the ‘rice of the petrochemical industry’ as it is used as a raw material for plastics, construction materials, vinyl, synthetic rubber, and more.
Ulsan National Institute of Science and Technology (UNIST) announced on the 24th that Professor Kwon Young-guk’s team from the Department of Energy and Chemical Engineering developed a copper-aluminum alloy catalyst that produces ethylene using carbon dioxide as a raw material.
The research team explained that when electricity is passed through an electrode coated with the catalyst, carbon dioxide undergoes an electrochemical reaction on the catalyst surface and is converted into ethylene.
The research was jointly conducted with Professor Lee Jong-hoon’s team from the Department of Materials Science and Engineering at UNIST, Professor Kim Woo-yeol’s team from KENTECH, and Professor Kim Hyung-joon’s team from KAIST.
The catalyst developed by the team attracted attention by achieving a current efficiency of 82.4%, the highest level reported in papers to date.
The current density (421 mA cm -2), which is used to assess the economic feasibility of commercializing the technology, also exceeded twice the evaluation standard. Higher catalyst efficiency means fewer by-products are generated.
Current density is an indicator that estimates the amount of ethylene that can be produced per unit time, and the minimum standard presented in technology economic evaluations is 200 mA cm -2.
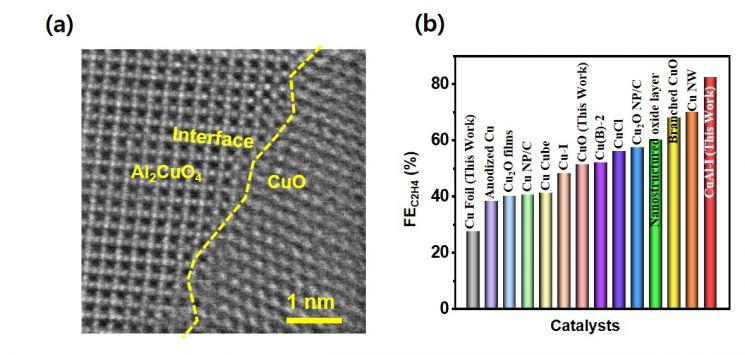
Electron microscope image of the developed catalyst (a) and catalyst performance graph (b). The efficiency of the developed catalyst (red bar on the right) is the highest.
원본보기 아이콘The method of synthesizing the catalyst is also simple, as it only requires co-precipitating copper and aluminum raw materials and then applying heat, making mass production easy.
Professor Kwon Young-guk said, “This is a win-win technology that removes greenhouse gases while producing ethylene in an eco-friendly way,” adding, “Since it meets the technology economic evaluation standards and the catalyst synthesis method is simple, it can contribute to accelerating the commercialization of eco-friendly ethylene production technology.”
The developed catalyst is in the form of copper oxide-aluminum (Al2CuO4) nanosheets uniformly coated with copper oxide (CuO) nanoparticles. The ethylene synthesis reaction is a multi-step process prone to by-product formation, and the two components of the catalyst share different stages of the catalytic reaction, allowing the ethylene synthesis reaction to occur quickly and abundantly.
Analysis showed that on the copper oxide surface, carbon dioxide (CO2) is converted into carbon monoxide (CO), while on the copper oxide-aluminum surface, carbon-carbon coupling reactions occur effectively.
The carbon-carbon coupling process is where the carbons of carbon monoxide (CO) meet to form the carbon bonds of ethylene (C2H4), known as the most challenging reaction. Copper oxide-aluminum also plays a role in effectively capturing activated carbon monoxide, maintaining a high concentration of the intermediate carbon monoxide, which facilitates the synthesis reaction.
Dr. Siraj Sultan from UNIST, along with researchers Lee Ho-jung from the Department of Energy and Chemical Engineering and Yoon Aram from the Department of Materials Science and Engineering, participated as first authors in this research. The results were first published online on March 22 in Energy & Environmental Science, a world-renowned journal in the energy and environmental science field, and are scheduled for formal publication.
The research was supported by the National Research Foundation of Korea’s Carbon to X technology development project, mid-career projects, and mid-career research projects.

(From the bottom left, counterclockwise) Researcher Lee Hojung (first author), Professor Kwon Youngguk, Dr. Shiraz Sultan (first author), Researcher Yoon Aram (first author), Choi Hansem, Researcher Gong Taehun.
원본보기 아이콘© The Asia Business Daily(www.asiae.co.kr). All rights reserved.