‘Cash-Rich’ SD Biosensor Expands Investment and Development
by Jang Hyowon
Published 07 Feb.2022 13:00(KST)
Updated 24 Jul.2023 20:03(KST)
The number of new COVID-19 cases is breaking records day after day. To respond to the spread of the Omicron variant, the government has shifted the COVID-19 diagnostic testing system to focus on self-diagnostic tests. The previously used polymerase chain reaction (PCR) tests are now prioritized for high-risk groups. There is growing anticipation that diagnostic companies such as SD Biosensor and Humasis, which have launched COVID-19 self-test kits, will benefit. Asia Economy examines the business structure, production capacity, and financial structure of SD Biosensor and Humasis to gauge their growth potential.
[Asia Economy Reporter Jang Hyowon] SD Biosensor is gaining renewed attention as the government’s diagnostic system changes due to the spread of the COVID-19 Omicron variant. This is because demand for COVID-19 self-diagnostic kits is expected to surge. Additionally, there is keen interest in what growth strategies SD Biosensor will pursue based on the cash reserves it has accumulated.
◆Q1 Earnings Outlook↑= SD Biosensor is an in vitro diagnostic specialist company established in 2010. It focuses particularly on immunochemical diagnostics, molecular diagnostics, point-of-care testing (POCT), and self-blood glucose monitoring. Until 2019, the self-blood glucose monitoring segment accounted for the largest share of sales, but after the COVID-19 pandemic in 2020, sales of immunochemical diagnostics surged dramatically, overwhelmingly increasing its share. As of the end of Q3 last year, the immunochemical diagnostics segment accounted for 91.5% of sales.
SD Biosensor owns the rapid immunodiagnostic brand ‘STANDARD Q’ within its immunochemical diagnostic product line, on which it based the development of its COVID-19 self-diagnostic kit.
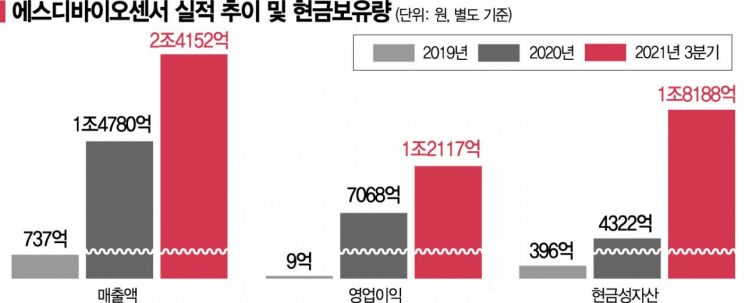
SD Biosensor’s performance made a quantum leap starting in 2020 as sales of COVID-19 diagnostic kits skyrocketed. In 2019, SD Biosensor’s sales and operating profit were KRW 73.7 billion and KRW 900 million, respectively, but in 2020, these figures rose to KRW 1.478 trillion and KRW 706.8 billion. Last year, sales are expected to reach KRW 2.9 trillion. This means sales increased 39-fold in just two years.
However, looking only at SD Biosensor’s Q4 results last year, performance is expected to fall short of market consensus. According to financial information provider FnGuide, the consensus estimates for SD Biosensor’s Q4 sales and operating profit last year were KRW 452.6 billion and KRW 188.3 billion, respectively. But three securities firms that issued reports on SD Biosensor this year estimate sales and operating profit to be between KRW 372.5 billion and KRW 399.9 billion, and KRW 152.3 billion to KRW 166.6 billion, respectively. All estimates are below consensus.
This is because the COVID-19 situation somewhat stabilized in Q4 last year, leading to a decline in demand for diagnostic kits. According to customs clearance data from Suwon City, where SD Biosensor is based, exports of immunochemical diagnostic kits in Q4 last year were USD 170 million, down 53% from the previous quarter. SD Biosensor generates 95.2% of its sales overseas in regions including Europe, Asia, and the Americas.
SD Biosensor’s performance, which had declined in Q4 last year, is expected to increase again following the recent spread of the Omicron variant. In December last year, SD Biosensor obtained Emergency Use Authorization (EUA) from the U.S. Food and Drug Administration (FDA) for its COVID-19 self-test kit and signed a supply contract worth approximately KRW 200 billion. It also plans to supply kits worth KRW 140 billion to Canada.
Furthermore, the South Korean government’s shift to a testing system centered on self-diagnosis and rapid antigen kits is another factor driving sales growth. After the government’s testing system transition, shortages of self-diagnostic kits have even appeared, indicating a surge in demand.
Lee Jisoo, a researcher at KTB Investment & Securities, said, “Due to the global resurgence of COVID-19, demand is increasing not only in North America but also in European and Asian countries, and domestic COVID testing methods are changing from PCR (polymerase chain reaction) to rapid antigen tests.” He analyzed, “As a result, sales and operating profit in Q1 this year are expected to increase by more than 160% compared to the previous quarter.”
◆KRW 1.8 Trillion in Cash Equivalents= Over the past two years, SD Biosensor has steadily accumulated cash reserves thanks to large-scale sales. As of the end of Q3 last year, SD Biosensor held approximately KRW 700 billion in cash. Including short-term financial products and financial assets, it has secured KRW 1.8 trillion in liquidity. Cash equivalents account for 68.7% of total assets.
With this massive cash reserve, SD Biosensor is engaging in mergers and acquisitions (M&A) and equity investments. In November last year, it acquired Eco Diagnostica, the second-largest diagnostic company in Brazil. SD Biosensor is using this company as a foothold to target the South American market. Local production also helps save taxes and allows allocation of domestic production quotas, making it advantageous.
Additionally, SD Biosensor made an equity investment in UXN, which is developing a platinum-based enzyme-free continuous glucose monitor. In September last year, SD Biosensor invested a total of KRW 40 billion in UXN’s third-party allotment capital increase, redeemable convertible preferred stocks (RCPS), and bonds with warrants (BW). The plan is to leverage UXN’s core technology and create synergy with SD Biosensor’s platform.
Won Jaehee, a researcher at Shinhan Investment Corp., analyzed, “Exports of COVID-19 rapid antigen kits and the inclusion of Brazil’s ECO company as a subsidiary will accelerate sales growth in the Americas.” He added, “Business diversification through M&A and a reduction in dependence on diagnostic kit sales are expected to bring valuation premiums.”
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.