Found Technology to Overcome Poor Absorption Drawback of 'Peptide' Drugs
Published 22 Jun.2021 08:54(KST)
GIST Professor Seo Ji-won’s Collaborative Research Team Uncovers Mechanism of Cyclic Peptide Drug Cell Membrane Penetration and Oral Administration Principles
[Asia Economy Reporter Kim Bong-su] Domestic researchers have elucidated the principles of cell membrane permeability and oral administration of cyclic peptide-type (amino acid cluster) drugs, presenting new possibilities for drug development.
The Gwangju Institute of Science and Technology (GIST) announced on the 22nd that a joint research team led by Professor Seo Ji-won from the Department of Chemistry identified the principles of cell membrane permeability and oral administration of cyclic peptides, solving the issues of low cell membrane permeability and low oral bioavailability?major drawbacks of peptides?and suggested the potential for developing orally administrable cyclic peptide-based new drugs.
Peptide drugs have long been overlooked in the pharmaceutical market due to many disadvantages. Although they effectively inhibit by mimicking active sites involved in protein-protein interactions, they suffer from low cell membrane permeability, poor oral bioavailability, and rapid degradation by peptide-degrading enzymes. However, advances in technologies such as mass production, sequence analysis, and drug formulation have begun to overcome these drawbacks. Currently, peptides account for about 5% of the global market, with an increasing number of approvals as drugs each year.
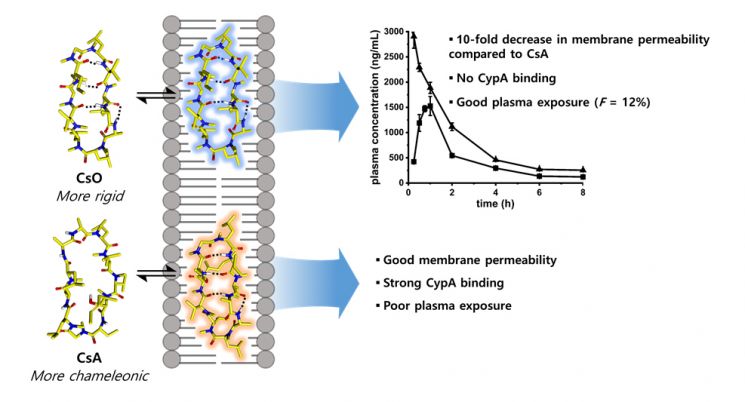
In particular, research interest is focused on cyclic peptides, where the C-terminus and N-terminus of peptides are linked to overcome peptide limitations. Cell membrane permeability and oral bioavailability are priority characteristics to improve for using cyclic peptides as drugs, and research is mainly based on naturally occurring cyclic peptides capable of cell membrane penetration. Notably, cyclosporin A, currently used as an immunosuppressant, is a representative orally administrable cyclic peptide drug attracting significant attention in studies on cell membrane permeability and oral administration.
The cyclic peptide discovered by the research team is cyclosporin O, a structural analog of cyclosporin A. Through comparative studies of the two substances, the team identified structural conditions necessary for cyclic peptides with molecular weights over 1000 Daltons (Da) to be developed as oral drugs.
The research team used two-dimensional nuclear magnetic resonance spectroscopy to confirm that cyclosporin A and cyclosporin O have similar structures under lipophilic conditions resembling the cell membrane environment but different structures under hydrophilic conditions. They also revealed that this difference affects the cell membrane permeability and oral bioavailability of cyclic peptides.
This property of changing structure depending on conditions is similar to how a chameleon changes its color according to its surroundings, hence it is called ‘chameleonicity.’ It is expected that regulating the chameleonicity of cyclic peptides will contribute to activating new drug development research with improved cell membrane permeability and oral bioavailability.
Professor Seo Ji-won stated, “We have presented important design principles for new drug development using cyclic peptides, a crucial molecular platform in mid-sized molecule drug development,” and added, “We plan to continue research on new drug development for infectious disease treatments such as antivirals using this cyclic peptide structure.”
The research results were published online on the 7th in the Journal of Medicinal Chemistry, an international journal in the field of medicinal chemistry published by the American Chemical Society.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.